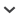We describe three Miocene species of Provanna from Japan, two new and one in open nomenclature, that represent the only known fossil examples from whale-falls and a considerable increase in the Miocene diversity of the genus. Provanna hirokoae sp. nov. comes from the latest Middle Miocene Kuroiwa seep site in central Honshu. The shells of this species are mostly recrystallized, but contain relict crossed lamellar microstructures. Provanna alexi sp. nov. is from the early Middle Miocene Shosanbetsu whale-fall site in northwestern Hokkaido, and has well preserved shells comprising an outer simple prismatic layer and an inner crossed lamellar layer. The two Provanna specimens from the Middle Miocene Rekifune whale-fall site, in eastern Hokkaido, are preserved as external moulds only, so are left in open nomenclature. Based on current knowledge, the presence of an outer prismatic layer and an underlying crossed lamellar layer seems to be a common feature in the shells of Provanna, as well as in other genera belonging to the family Provannidae and the superfamily Abyssochrysoidea. Although the oldest occurrence of Provanna was in the Late Cretaceous, the genus did not spread geographically and ecologically until the Miocene (with four, or possibly five species), a date concordant with some molecular estimates. However, this could be an artefact of the fossil record because the known pre-Miocene seep and whale-falls are more geographically restricted than those from the Miocene.
Introduction
The small gastropod genus Provanna Dall, 1918 is one of the characteristic and species-rich molluscan taxon in modern chemosynthetic faunas (e.g., Warén and Bouchet 1993,2001, 2009; Sasaki et al. 2010). Most Provanna species are grazers on filamentous bacteria, while some are detritus feeders. Although Bergquist et al. (2007) suggested that Provanna variabilis Warén and Bouchet, 1986 may also harbor symbiotic bacteria, Sasaki et al. (2010) raised doubts about this interpretation based on the anatomy of the species.
Eighteen Recent species of Provanna have so far been described (Table 1), most from hydrothermal vent and hydrocarbon (cold) seep sites, but some also from whale-fall and wood-fall sites (e.g., Smith and Baco 2003; Warén and Bouchet 2001,2009; Sasaki et al. 2010). In contrast to this diversity, only four fossil Provanna species have been formally described: two from Cretaceous seep deposits in Hokkaido, Japan, one from upper Eocene to Oligocene seep deposits and upper Eocene wood-falls in Washington State, USA, and one from Miocene seeps in New Zealand (Table 1). In addition to these, fossil gastropods from four different Miocene localities have been figured, but not formally described, as Provanna or other provannids. These include “provannids” from the Freeman's Bay Limestone (a suspected seep deposit) of the Miocene Lengua Formation, Trinidad (Gill et al. 2005: fig. 7I, J) and three Provanna species from two whalefall sites and one seep site from Japan (Amano and Little 2005; Amano et al. 2007, 2010). Here we formally describe these Japanese species and discuss the shell microstructures and the fossil record of the genus Provanna. The Japanese occurrences represent the only known fossil examples of Provanna from whale-falls and a significant increase in the Miocene species diversity of the genus.
Institutional abbreviations.—JUE, Joetsu University of Education, Joetsu, Japan.
Material
Twenty-seven Provanna specimens were collected from the Shosanbetsu whale-fall site in northwestern Hokkaido (locality 1 in Fig. 1; Amano and Little 2005). This site is from the lower Middle Miocene (15.9–14.9 Ma) Chikubetsu Formation (Amano et al. 2007). We examined and measured nine of the specimens, all of which have well preserved shells, allowing microstructural details to be studied.
Two Provanna specimens were collected from the Rekifune whale-fall site in eastern Hokkaido (locality 2 in Fig. 1; Amano et al. 2007). This site is from the Middle Miocene Nupinai Formation (ca. 13-12 Ma). The specimens are preserved as external moulds only, from which silicone rubber casts were made for examination.
A total of 124 Provanna specimens were collected from a hydrocarbon seep deposit at the Kita-Kuroiwa Quarry in Kakizaki-ku, Joetsu City, Niigata Prefecture, central Honshu, Japan (locality 3 in Fig. 1; Amano et al. 2010). The Kuroiwa seep site is from the uppermost Middle Miocene (11.64±0.65 Ma) Ogaya Formation (Amano et al. 2010). Twenty-eight of the specimens are relatively well preserved and were examined in detail. The shells of these specimens are recrystallized, but relict microstructural details are visible in a few.
To compare shell microstructural details of our fossil Provanna species with modern species we studied the shell of one specimen of P. reticulata Warén and Bouchet, 2009. This was collected with samples of seep carbonate (M56B, GeoB 8212-1 TV grab) from 3100 m water depth at the Hydrate Hole pockmark site, on the Congo deep-sea fan (Sahling et al. 2008) during RV METEOR Cruise M56 (Chief Scientist Gerhard Bohrmann, MARUM, University of Bremen, Germany).
Systematic paleontology
Phyllum Mollusca Linnaeus, 1758
Class Gastropoda Cuvier, 1797
Order Caenogastropoda Cox, 1959
Superfamily Abyssochrysoidea Tomlin, 1927
Family Provannidae Warén and Ponder, 1991
Genus Provanna Dall, 1918
Type species: Trichotropis (Provanna) lomana Dall, 1918; Recent, US Pacific Coast.
Provanna hirokoae sp. nov.
2010 Provanna sp.; Amano et al. 2010: figs. 5C—E.
Etymology. Named for the first author's wife Hiroko.
Type material: Holotype, JUE 15901 (Fig. 2A); Paratypes, JUE 15902-1 (Fig. 2B), JUE 15902-2 (Fig. 2C), JUE 15902-3 (Fig. 2D), JUE 15902-4 (Fig. 2E).
Type locality. Kita-Kuroiwa Quarry, Kakizaki-ku, Joetsu City in central Honshu, Japan (see Amano et al. 2010); seep site.
Type horizon: Fossil hydrocarbon seep deposits of the Ogaya Formation, uppermost Middle Miocene.
Dimensions.—See Table 2.
Diagnosis.—Medium-sized Provanna with variable sculpture ranging from sigmoidal axial growth lines and many indistinct spiral cords to strong sigmoidal axial ribs crossed by weaker spiral cords. Whorls with broad, smooth sutural ramp and distinct rounded shoulder, tabulated at axial ribs, where present.
Description.—Medium-sized shell, up to 9.8 mm high, ovate fusiform; teleoconch at least three whorls; protoconch not preserved. Sutures weakly impressed. Body whorl with broad, smooth sutural ramp sloping gently to rounded shoulder. From shoulder abapically body whorl sides gently curved. Ornament variable; some specimens, like holotype (Fig. 2A) and paratype JUE 15902-2 (Fig. 2C), having whorls with sigmoidal growth lines and many indistinct spiral cords best developed on the body whorl near the shoulder and at whorl base; other specimens, like paratype JUE 15902-3 (Fig. 2D) having nearly smooth whorls with sigmoidal growth lines only. Strong sigmoidal axial ribs on apical whorls of some specimens, such as paratype, JUE 15902-4, up to eighteen in number on penultimate whorl (Fig. 2E). In some specimens, distinct axial ribs also on body whorl, as in paratype, JUE 15902-1 (Fig. 2B). In specimens with axial ribs, spiral cord at shoulder at deflection point in shape of ribs relatively stronger than other spiral cords, forming weak nodes at intersection points with axial ribs. Aperture broad and rounded, with evidence for weak siphonal notch. Shells mostly recrystallized, but relict crossed lamellar structure visible on inner shell surface of one specimen.
Remarks.—Provanna lomana Dall, 1918 from seep, vent, and whale-fall sites off California resembles P. hirokoae in outline, shell size and shape of axial ribs on the apical whorls. However, P. lomana has stronger spiral cords and axial ribs on the body whorl. P. chevalieri Warén and Bouchet, 2009 from seep sites off West Africa is also similar to P. hirokoae in outline and size, but differs in having deeper sutures, a narrower and less sloping sutural ramp, and strong spiral cords on the basal part of the body whorl.
Provanna hirokoae specimens have very variable sculpture, particularly on the early whorls, and share this characteristic with several fossil (P. antiqua Squires, 1995 and P. marshalli Saether, Little, and Campbell, 2010) and modern (P. variabilis) species in the genus.
Stratigraphic and geographic range.—From the type locality only.
Table 1.
Distribution, ecology, and shell microstructure of the fossil and modern species of Provanna. 1) after Kiel (2010). The fossil occurrence bathymetry estimations are in brackets because they are estimations. 2) The type material for these species was trawled and therefore the original habitats are unknown. The habitats given are from subsequent discoveries. 3) From inner layer (left) to outer layer (right); ccl, complex crossed lamellar structure; cl, crossed lamellar structure; homo, homogeneous structure; spl, simple prismatic structure.

Table 2.
Measurements of Provanna hirokoae sp. nov. specimens.

Table 3.
Measurements of Provanna alexi sp. nov. specimens.

Provanna alexi sp. nov.
2005 Provanna sp.; Amano and Little 2005: figs. 5H—K.
Etymology. Named for the second author's son Alex.
Type material: Holotype, JUE 15904 (Fig. 3A); Paratypes, JUE 15905-1 (Fig. 3B), JUE 15905-2 (Fig. 3C), JUE 15905-3 (Fig. 3D).
Type locality. 5.5 km upstream of Shosanbetsu Village on the Setakinai River in northwestern Hokkaido, Japan (see Amano and Little 2005); whale-fall site.
Type horizon: Fossil whale-fall deposits of the Chikubetsu Formation, lower Middle Miocene.
Dimensions.—See Table 3.
Diagnosis.—Small-sized Provanna with strong, tabulated shoulder, 8–16 spiral cords and 22–26 axial ribs on body whorl.
Description.—Shell small, up to 6.5 mm high, with ovate fusiform shape; teleoconch with three whorls; protoconch not preserved. Sutures weakly impressed. Apical whorls sculptured by strong spiral cords, three to six in number on penultimate whorl, and strong, straight axial ribs, six to twenty-one in number on penultimate whorl. Very distinct angular shoulder formed by strong spiral cord with strong nodes at intersection with axial ribs. Narrow sutural ramp adapically of shoulder. Whorl sides ranging in shape from nearly straight to weakly curved. Stronger spiral cords causing distinct angulations in body whorl sides of some specimens. Body whorl large, comprising roughly 60–70% of shell height. Ornamentation on body whorl weaker than on apical whorls, with eight to sixteen spiral cords, best developed near shoulder and whorl base, and twenty-two to twenty-six axial ribs. Shoulder also weaker on body whorl and sutural ramp having greater slope angle. Aperture ovate with suggestion of weak apertural notch. Shells formed of two distinct microstructural elements: inner crossed lamellar layer (up to 120 µm thick) and outer simple prismatic layer (up to 20 µm thick).
Remarks.—In having a tabulate shoulder, Provanna alexi is similar to P. antiqua from Eocene wood-falls and Eocene to Oligocene seep deposits in Washington State, USA and P. marshalli from Miocene seeps of New Zealand. However, P. antiqua differs from P. alexi by having less numerous spiral cords on the body whorl (2–7 in P. antiqua; 8–16 in P. alexi). The number of spiral cords in P. marshalli (0–11) is generally less than in P. alexi, but there is some overlap. In addition, axial ribs are always present in P. alexi, whereas this character is lacking in some specimens of both P. antiqua and P. marshalli.
Stratigraphic and geographic range.—From the type locality only.
Fig. 2.
Abyssochrysoid gastropod Provanna hirokoae sp. nov., uppermost Middle Miocene Ogaya Formation (locality 3), Japan. A. Holotype, JUE 15901; apertural (A1), adapertural (A2), and apical (A3) views. B. Paratype, JUE 15902-1; adapertural (B1), oblique adapertural (B2), and apical (B3) views. C. Paratype, JUE 15902-2, apertural view. D. Paratype, JUE 15902-3; adapertural (D1) and apical (D2) views. E. Paratype, JUE 15902-4; apertural (E1), adapertural (E2), and apical (E3) views.

Provanna sp.
2007 Provanna? sp.; Amano et al. 2007: fig. 3A, B.
Material.—Two specimens from the whale-fall site of the Middle Miocene Nupinai Formation, eastern Hokkaido, Japan.
Measurements.—See Table 4.
Description.—Shell small, up to 5.0 mm high, ovate fusiform; teleoconch with three whorls; protoconch not preserved. Suture very shallow; whorl side curved without shoulder. Surface ornamented by distinct axial ribs and spiral cords, forming cancellate sculpture at crossing points. Axial ribs almost straight, numbering 13 on penultimate whorl and 16 on body whorl. Four spiral cords on penultimate whorl and eight on body whorl.
Remarks.—There are three Provanna species with cancellate ornament that can be compared to the Rekifune species. However, two of these (P. admetoides Warén and Ponder, 1991 and P. marshalli Saether, Little, and Campbell, 2010) have distinct tabulated shoulders, lacking in the Rekifune species, and one (P. nassariaeformis Okutani, 1990) has a wider body whorl than seen in either of the Rekifune specimens.
We do not name this species at present because we have only two specimens, and these are present as external moulds only. Amano et al. (2007) illustrated these specimens as Provanna? sp., but small gastropods having such cancellate sculpture recorded from chemosynthetic communities can be also be found amongst the nassariids, cancellariids, and turrids. However, we are confident that the Rekifune specimens do not belong to these families for the following reasons. The lack of a fasciole and folding on the aperture inner lip suggests the Rekifune specimens are not nassariids, and collumellar folds, which are characteristic of the cancellariids, are also lacking in the Rekifune specimens, indicating they do not belong to this family either. Further, species of the nassariids and cancellariid are rare in the modern chemosynthetic fauna. The Rekifune specimens lack the posteriorly situated growth lines which characterize the turrids.
Table 4.
Measurements of Provanna sp. specimens.

Fig. 3.
Abyssochrysoid gastropod Provanna alexi sp. nov., lower Middle Miocene Chikubetsu Formation (locality 1), Japan. A. Holotype, JUE 15904; apertural (A1), adapertural (A2), oblique adapertural (A3), and apical (A4) views. B. Paratype, JUE 15905-1; apertural (B1), oblique adapertural (B2), adapertural (B3), and apical (B4) views. C. Paratype, JUE 15905-2; apertural (C1) and apical (C2) views. D. Paratype, JUE 15905-3; apertural (D1) and apical (D2) views.

Fig. 4.
Abyssochrysoid gastropod Provanna sp., Middle Miocene Nupinai Formation (locality 2). Silicone rubber moulds coated with ammonium chloride. A. JUE 15846-1, apertural view. B. JUE 15846-2, adapertural oblique view.

Fig. 5.
Shell microstructure of fossil and modern species of Provanna. A. Recent Provanna reticulata Warén and Bouchet from Hydrate Hole pockmark, Congo Fan, West Africa (M56B, GeoB 8212-1 TVG), JUE 15907; top of aperture where it meets the body whorl wall (A1), outer lip of aperture (A2). B—D. Provanna alexi sp. nov. B. Holotype, JUE 15904, top of aperture where it meets the body whorl wall. C. Topotype, JUE 15906-1, fractured apical whorl shell, white arrows indicate relict crossed lamellar structures in the innermost layer. D. Paratype, JUE 15905-1, fractured apical whorl shell. E. Provanna hirokoae sp. nov., paratype, 15902-4, fractured body whorl shell. Abbreviations: cl, crossed lamellar layer; ?cl, possible crossed lamellar layer; p, periostracum; spl, simple prismatic layer.

Discussion
Shell microstructures in Provanna .—In contrast to the numerous published studies on the anatomy and gross shell morphology of Provanna species there are few reports detailing shell microstructures, within the genus specifically, and the provannids more widely (Table 1). Kiel (2004) showed that the shell of P. variabilis Warén and Bouchet, 1986 has three layers of microstructure: an outer thin simple prismatic layer, a thicker central complex crossed lamellar layer and a thin inner simple prismatic layer (Table 1). He noted that the innermost part of the complex crossed lamellar layer sometimes has loosely packed crystals (Kiel 2004: fig. 30) and the inner prismatic layer is not present in all parts of the shell (Kiel 2004: fig. 32). Kiel (2004) identified the same microstructural shell composition in the large provannid Alviniconcha hessleri Okutani and Ohta, 1988, but in this species the layers are extremely thin compared to the thick outer periostracum (Kiel 2004: fig. 33).
We found the same layers in the modern species P. reticulata (Fig. 5A1, A2). Around the fractured aperture of the studied specimen the outer prismatic layer stays roughly the same width (ca. 20 µm), although it slightly thickens (to 25 µm) where the spiral cords intersect with the shell edge. The inner prismatic layer is present along the whole aperture margin, but thickens substantially (to ca. 50 µm) adapically where the outer lip of the aperture meets the body whorl wall (Fig. 5A1). Both the outer simple prismatic and crossed lamellar layers are present in the shells of the fossil species P. alexi sp. nov. and P. marshalli Saether, Little, and Campbell, 2010 (Table 1). In P. alexi these two layers are very clear towards the exterior of the shell (Fig. 5B), but there is also an inner layer of variable thickness, sharply delineated from the crossed lamellar layer, formed mostly of small crystals without obvious orientation (Fig. 5C, D). This layer also often has vertical structures running through it, and, in places, relict patches of crystals with oblique orientation which are parallel to the crystals in the crossed lamellar layer (Fig. 5C). We suggest that this layer is the innermost portion of the crossed lamellar layer that is undergoing dissolution and recystallization. It might correspond to the inner zone of loosely packed crystals in the crossed lamellar layer in P. variabilis (as seen in Kiel 2004: fig. 30). There is no inner prismatic layer in P. alexi, which is also the case for P. marshalli. Saether et al. (2010) suggested that the lack of this layer in P. marshalli might have been due to differential taphonomic and diagenetic processes affecting the shells, either on the seafloor or during early burial, where the outer shell microstructural layers were protected for longer from these processes by the periostracum in relation to the innermost shell layers. An alternative explanation is that an inner layer of simple prismatic microstructure was not present in P. alexi and P. marshalli. The shells of P. hirokoae sp. nov. are largely recrystallized, but relict textures in the crystals on the inner surface of one specimen (Fig. 5E) indicates that crossed lamellar microstructures were present in this species. Kiel (2006) identified three microstructural layers in the shell of the fossil species P. antiqua Squires, 1995: an outer prismatic layer, a central complex crossed lamellar layer and an inner homogeneous layer. Given what we have seen in P. alexi this inner layer of P. antiqua might not be a primary structure, but could rather represent an innermost layer of dissolution and recrystallization, as suggested for P. marshalli by Saether et al. (2010).
Amongst gastropods related to the provannids shell microstructures have been recorded in the fossil abyssochrysoidean gastropod Hokkaidoconcha hikidai Kaim, Jenkins, and Warén, 2008 from the Cretaceous seep sites in Hokkaido. This species has an outer prismatic layer, a crossed lamellar central layer and an inner prismatic layer (Kaim et al. 2009: fig. 11B3). So, based on the available data, it seems that the presence of an outer prismatic microstructural layer and an underlying crossed lamellar microstructural layer is a common feature in the shells of all Provanna species, as well as in other provannid genera and in the superfamily Abyssochrysoidea. The structure of the innermost layer, where present, may be more variable (simple prismatic or homogeneous), although identification of microstructures is more difficult in fossil specimens where shell preservation is an issue.
Fossil record of Provanna .—The oldest record of the genus is P. tappuensis Kaim, Jenkins, and Warén, 2008 from a Middle Cenomanian (Late Cretaceous, 95–96 Ma) seep site in Hokkaido, where it co-occurs with another provannid species, Desbruyeresia kanajirisawensis Kaim, Jenkins, and Warén, 2008, and a hokkaidoconchid species, Hokkaidoconcha tanabei Kaim, Jenkins, and Warén, 2008 (Kaim et al. 2008). The only other Mesozoic record of Provanna is P. nakagawaensis Kaim, Jenkins, and Hikida, 2009 from two Lower Campanian (Late Cretaceous, 80–83 Ma) seep sites and one Coniacian (86–89 Ma) wood-fall site, also in Hokkaido (Kaim et al. 2009). This species co-occurs Hokkaidoconcha hikidai at one of these sites (Kaim et al. 2009).
In the Paleogene and Neogene the range of habitats and geographic areas from which fossil Provanna specimens have been found increases in relation to the Mesozoic occurrences (Table 1). In the Paleogene P. antiqua is found in five Late Eocene to Late Oligocene (23–37 Ma) seep sites and also in two Late Eocene (34–37 Ma) wood-fall sites in Washington State, USA (Goedert and Campbell 1995; Squires 1995; Squires and Goedert 1995; Rigby and Goedert 1996; Peckmann et al. 2002; Kiel 2006; Kiel and Goedert 2006a, b). In the Early to Middle Miocene Provanna species are found in seep sites in New Zealand (P. marshalli), Japan (P. hirokoae) (Table 1), and possibly also Trinidad (Gill et al. 2005; Saether et al. 2010). Also in the Miocene two species of Provanna (P. alexi and Provanna sp.) occurred at whale-falls. Today only one Provanna species (P. lomana), is known from modern whale-fall communities (Smith and Baco 2003), although two species of the provannid genus Rubyspira occur at whale-falls in Monterey Bay, California (Johnson et al. 2010). The modest species diversification of Provanna in the Early to Middle Miocene has some congruence with the upper estimates in Johnson et al. (2010) for the radiation of the genus at 15–35 Ma (vicariance calibration) or 17–45 Ma (fossil calibrations). However, it should be noted that for at least two of the Miocene occurrences of Provanna or provannids (New Zealand and Trinidad) there are presently no known older seep deposits in the same geographic areas, meaning that there could have been a greater pre-Miocene diversity and palaeobiogeographic distribution in Provanna, not currently seen in the fossil record. This alternative would be more consistent with the older molecular divergence estimates in Johnson et al. (2010).
Acknowledgements
We thank Misaki Aikawa (Okawa City Office, Japan) for helping us collect specimens of Provanna hirokoae. We also thank Gerhard Bohrmann and Jörn Peckmann (both, MARUM, University of Bremen, Germany) for specimens of Provanna reticulata from the Congo Fan seeps, and Steffen Kiel (University of Göttingen, Germany) for his information about the shell microstructure of Provanna. Useful comments from Anders Warén (Swedish Museum of Natural History, Sweden) and an anonymous referee are acknowledged. This study was partly supported by a Grant-in-aid for Scientific Research from the Japan Society of Promotion of Science (C, 23540546, 2011–2013) to KA, and a Hanse-Wissenschaftskolleg Fellowship to CTSL.