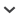Fossil material from the lower Miocene collected in the basin lake of Beşkonak (Turkey) included 19 slabs showing 19 amphibian anuran tadpoles of rather large size, at Gosner stages 36–38. These well preserved specimens show many morphological and skeletal characters. They are here tentatively referred to the genus Pelobates. Two of these tadpoles show an unusual group of black roundish spots in the abdominal region, and a third similar group of spots is present in another slab but we were unable to state if it was associated with a tadpole or not. Several hypotheses can be proposed to account for these structures: artefacts; intestinal content (seeds; inert, bacterial or fungal aggregations; eggs); internal or external parasites; diseases; eggs produced by the tadpole. The latter hypothesis is discussed in detail and is shown to be unlikely for several reasons. However, in the improbable case where these spots would correspond to eggs, this would be the first reported case of natural paedogenesis in anurans, a phenomenon which has been so far considered impossible mostly for anatomical reasons (e.g., absence of space in the abdominal cavity).
Introduction
Because they are composed of about 90% water (Bradford 1984), tadpoles of anuran amphibians are extremely fragile organisms: just a few hours after having been taken out of water and deposited on the ground, virtually nothing remains of them. Therefore tadpoles, although periodically very abundant in some continental aquatic habitats, are not good candidates for fossilisation, and only relatively few fossil tadpoles are known, presumably having benefited from unusual fossilisation conditions. Among the best preserved ones are the lower middle Miocene fossil tadpoles from two localities of the Gürcü Valley (40°35–38′N, 32°39–42′E, altitude 1200–1300 m), about 90 km north of Ankara, Turkey, described and illustrated by Wassersug and Wake (1995), and those from a nearby locality, the basin lake of Beşkonak (40°38′N, 32°41′E), also situated in the Gürcü Valley, reported by Rückert-Ülkümen et al. (2002). In both cases, the tadpoles were referred to the genus Pelobates Wagler, 1830 (Pelobatidae Bonaparte, 1850). The age of the fossil deposits of the Gürcü Valley was estimated to 14.1 ± 1 Mya, corresponding to the middle Miocene, dated using K/Ar isotopes of biotites contained in the rocks (Kasapligil 1981), but this date was challenged by subsequent studies of the flora and of the insect fauna (Paicheler 1988; Nel and Paicheler 1993), which suggest that these deposits are from the Oligocene-Miocene boundary or the lower Miocene.
We here report on several well preserved anuran tadpoles also from the basin lake of Beşkonak (Fig. 1). These specimens were collected by one of us (JCP) in 1972, but never described in detail, although they were briefly mentioned in a general paper dealing with the fossils of this locality (Paicheler et al. 1978). The fossilisation occurred as a result of volcanic activity which poured lava and hot volcanic ash into the lake, causing rapid sedimentation of diatomaceous frustules. These, along with the accumulation of fine silt from streams flowing into the lake, formed the laminated diatomites and paper shales (Kasapligil 1981). The excellent preservation of the animals also suggests reducing conditions on the bottom of the lake with a H2S layer (Paicheler et al. 1978).
The fauna and flora found with these tadpoles are all aquatic organisms or organisms linked to aquatic habitats. This area was therefore presumably flooded at the time of fossilisation. According to the floristic, palynologic and entomological analyses, the regional climate was hot temperate of a Mediterranean type (Paicheler et al. 2007). This area was subject to regular alternation of dry and wet seasons. Quite shallow, the lake presumably varied from a great lake connected with other basins to a huge swamp, depending on the season (Paicheler et al. 1978).
Fig. 1.
Map of Northwestern Turkey showing the area which yielded the fossil tadpoles in this study, near Güvem, in the Gürcü Valley. See Fig. 2 for detailed map of this area.

The tadpoles appear as organic film imprints within a diatomite matrix. They are well preserved, lying on their side, on their venter or ventrolaterally and thus showing details of their soft anatomy, such as gills, eyes, lungs, parts of the intestinal tract and blood vessels, caudal myotomes and caudal fins. Bones and cartilage present some depressions. The slabs that contain the tadpoles also yield plant remains and a tiny bird feather, but no adult amphibians. However the most striking feature of these specimens is that two of them show the clear presence of rounded spots in the abdominal region. Though there are several alternate explanations, the question arises whether these spots can be interpreted as eggs produced by the tadpoles.
Material and methods
We examined traces of fossil tadpoles from 18 distinct slabs over several thousand slabs containing tadpoles observed in the field in this region in 1972 by JCP. These slabs with tadpoles were collected in two different localities of this basin: Ahlath Dere, north of the village of Kerimler; and west of the village of Agaöz (Fig. 2). Both localities are from the same stratigraphic level (Paicheler 1988). We identified 19 individual tadpoles in these slabs. These slabs, as well as another one showing a group of roundish spots, discussed below, are housed in the Paris Museum collection (Reptiles and Amphibiens), under the numbers MNHN 1998.9173–9194.
The tadpoles were observed under a binocular Leica MS5 and measured with a hand calliper due to their large size. Based on their cranial ossification, they were tentatively referred to the standard developmental stages of the table of Gosner (1960).
Description of the material
The tadpoles.—The 19 tadpoles are well preserved and do not show distortions or crushing. They are similar in aspect, without any trace of posterior limb buds, thus suggesting that they were in rather early stages of larval development, before the beginning of the period of metamorphosis. However, they show a somewhat extended range of sizes, so that it can be hypothesized that they were not all of the same age, and therefore originated from different egg clutches, laid at different dates. The absence of hindlimb traces precludes their assignation to an accurate developmental stage. However, some indices could help to determine a range of developmental stages (see below).
Fig. 2.
Sites of collection of the fossil tadpoles in the Gürcü Valley studied here (enlarged from Fig. 1), Turkey. A. Ahlath Dere, where the specimen MNHN 1998.9173 was collected. B. Agaöz, where the specimen MNHN 1998.9174 and the slab with the mass of roundish spots MNHN 1998.9191 were collected.

Anteriorly, the keratinised beaks are visible in a few specimens, as well as the serrations in one of them. The upper beak seems to have the form of a large arch and the lower beak of a broad “U”. Keratodonts have not been observed.
Skeletal elements are visible in almost all tadpoles, especially the frontoparietal and the vertebrae, although the parasphenoid and the otic capsules are also visible in a few. The frontoparietal bones lie laterally to the cultriform process of the parasphenoid; they are elongate, drop-shaped, tapering anteriorly and enlarged posteriorly to a blunt extremity. The lateral notches present on the frontoparietal of the specimen UCMP 139183, described in detail by Wassersug and Wake (1995), are absent in all of our specimens exhibiting frontoparietal bones. However, these specimens exhibit a small concavity which could correspond to the fontanelle. The parasphenoid is well visible in one specimen. It has the form of an inverted T, with the cultriform process long and fine and ending in a point anteriorly. The alar processes are fine triangles with a pointed tip. The posterior end of the parasphenoid forms an obtuse angle.
Imprints of otic capsules are present in some fossils. They are rounded structures situated between the frontoparietals and the alar processes of the parasphenoid and lateral to them. They are always smaller than the eyes, from about half size to almost similar size.
Between eight and eleven vertebrae are observable in these specimens. The first vertebra, the atlas, is not well outlined but seems thicker than the following ones, although what we observed could be a print of the atlas and of the first post-atlas vertebra. Each vertebra bears well developed transverse processes, the diapophyses. The diapophyses of the three first vertebrae are straight, the following narrowing in their middle, then widening in their distal part. Furthermore, their length decreases in an anteroposterior direction along the vertebral series although the size of all vertebrae is equal. The last vertebra is smaller than the others in one specimen. Along the vertebra series, the imprint of the notochord is often visible.
The absence of hindlimb traces suggests that these tadpoles are in a rather early developmental stage, at least before stage 30 in which the hindlimb buds are rather developed (Gosner 1960). However, the ossification of skeletal elements described above gives some contradictory indication upon their developmental stage. In several families of living anurans, the first three osseous structures that appear during the ossification of the skull are the otic capsules, the parasphenoid and the frontoparietals, which develop during the stages 36–38 and are present at the latest at the end of stage 38 (Kemp and Hoyt 1969; Gaudin 1973, 1978; Hanken and Hall 1984, 1988; de Sá 1988; Wiens 1989; Trueb and Hanken 1993; Maglia and Púgener 1998). We therefore conclude that the Turkish tadpoles are in developmental stages 36–38. The same interpretation was proposed by Wassersug and Wake (1995) for their tadpoles, although in this case also no hindlimb structures were visible. In fact, we know of no instance of fossil tadpoles at these stages showing hindlimbs: among the many photographs of tertiary tadpoles shown in the book by Špinar (1972), the only ones that show hindlimbs are at later stages, just before the end of metamorphosis.
The abdominal roundish spots.—Among the 19 tadpoles examined, two show a very striking peculiarity, i.e., the presence of roundish dark spots in the abdominal region.
The first specimen (MNHN 1998.9173, BL 37.2 mm), collected in locality (A) Ahlath Dere (Fig. 2), shows about 100 such spots concentrated in the posteroventral part of its abdomen, a part of the tadpole's body which is usually entirely filled up with the intestine (Fig. 3). These spots are all of similar aspect and size, very regular in shape, roundish and with a diameter of 0.76–1.06 mm (n = 7). They are as dark as the eye imprints of the tadpoles, and darker than the remains of other organs and soft parts, suggesting either a protein-rich structure or more probably that they are due to a bacterially induced pyritization (Briggs 2003). Some of these spots appear isolated, whereas others appear in close contact or even connected to each other. The abdomen of this specimen has a fully normal shape, not being particularly expanded or deformed by the presence of these structures.
The second specimen (MNHN 1998.9174, BL 36.6 mm), collected in locality (B) Agaöz (Fig. 2), shows only a few spots (roughly 13), with a similar shape and slightly larger (diameter 0.91–1.37 mm; n = 4) than in the first specimen (Fig. 4). These spots are located ventrally and in the posterodorsal part of the abdomen, which also has a normal shape.
Finally, on a slab (MNHN 1998.9191) also collected in Agaöz, a third group of similar spots is present but as all the surrounding (and a part of this group of spots) is missing, we are unable to state if it was also present within a tadpole or not (Fig. 5). The number of spots is estimated to more than 150 and their mean diameter is 1.3 mm (1.0–1.6 mm; n = 8). The aspect and size of these spots suggest that they are of the same nature as those observed in the two tadpoles.
Unfortunately, the preservation of these soft structures as a film rather than as three dimensional structures prevents us from extracting more information from these spots than the observation of these structures, and hypotheses have to be proposed to explain their presence.
Whatever the origin of these spots is, it is worth noting that none of the 218 slabs bearing fossil actinopterygian Cyprinidae Cuvier, 1816 (all referred to Leuciscus Cuvier, 1816) collected with these fossil tadpoles, presents such structures (Gaël Clément, personal communication 2008).
Discussion
Identification of the tadpoles.—Taxonomic identification of these tadpoles is difficult. No specimen shows well-preserved mouthparts with keratodont rows that would allow their straightforward allocation to a genus. The large size of some of these tadpoles (about 90 mm; see Table 1) excludes a number of extant anuran genera, but leaves several possibilities. Tadpoles of the genera Alytes Wagler, 1829 (Alytidae Fitzinger, 1843), Pelobates (Pelobatidae), Pelodytes Bonaparte, 1838 (Pelodytidae Bonaparte, 1850) and Pelophylax Fitzinger, 1843 (Ranidae Rafinesque-Schmaltz, 1814) are known to reach similar large sizes: Alytes obstetricans (Laurenti, 1768), 90 mm (Boulenger 1891); Pelobates fuscus (Laurenti, 1768), 200 mm (Grillitsch et al. 1983); Pelobates syriacus Boettger, 1889, 205 mm (Boschwitz 1957); Pelodytes punctatus (Daudin, 1802), 95 mm (van den Elzen 1976); Pelophylax esculentus (Linnaeus, 1758), 163 mm (Borkin et al. 1982); Pelophylax ridibundus (Pallas, 1771), 165 mm (Borkin et al. 1982); and Pelophylax perezi (Seoane, 1885), 90 mm (Llorente et al. 1995). All these tadpoles are usually smaller than the sizes just listed, but can reach them when they overwinter without metamorphosing (Boulenger 1891; van den Elzen 1976; Savage 1952; Grossenbacher 1997; Kuzmin 1997; Miaud and Muratet 2004). In normal conditions however, the species of the genus Pelobates are those which reach the largest size before metamorphosis among western Palearctic frog tadpoles (Díaz-Paniagua 1988). Tadpoles without a thyroid gland or with a deficiency in the hormonal system (axis hypothalamus-hypophysis-thyroid) are known to reach sometimes unusual large sizes, e.g., four times longer than in normal tadpoles in Xenopus laevis (Daudin, 1802) (Boschwitz 1957; Borkin et al. 1982; Rot-Nikcevic and Wassersug 2004), but this does not seem to be the case in our fossil tadpoles (see Suggestions below).
Fig. 3.
Photograph (A) and drawing (B) of the fossil tadpole MNHN 1998.9173 from Ahlath Dere, Gürcü Valley, lower Miocene, Turkey, bearing a group of spots in the abdominal region.

Table 1.
Some measurements (in millimetres) of fossil tadpoles at Gosner (1960) stages 36–38 from the basin lake of Beşkonak. Only the 6 most complete tadpoles were measured.

Palaeontological and distributional data also allow some inferences. The continental Miocene of Turkey is marked by major faunal interchanges between Africa and Laurasia, so the fossil tadpoles can also have a north African origin, and the anuran genera present in this region are considered here as well as the native fauna from Turkey. The genus Alytes can rather safely be excluded, as it is currently know only west of the Alps and in northern Africa, but no fossil of this genus is known from the eastern Mediterranean region. The other three groups are currently represented in the native fauna of Turkey (Basoglu and Özeti 1973; Baran and Atatür 1998), so that they can remain potential candidates. Fossils of the family Pelobatidae are known since the early Eocene (MP 7) for Eopelobates Parker, 1929 and the early—middle Oligocene (MP 22 or 23, 30–31 Mya) for Pelobates (Rage and Roček 2003). Fossils of Pelodytidae are known since the late Eocene (MP 16) or perhaps middle Eocene (MP 13, about 40 My a), and since the middle Miocene (11–12 Mya) for Pelodytes although this genus is much older (Rage and Roček 2003). Finally, green frogs of the genus Pelophylax (Frost et al. 2006) are known since the early Oligocene (MP 22, 32–33 Mya) (Rage and Roček 2003).
Fig. 4.
Photograph of the fossil tadpole MNHN 1998.9174 from Agaöz, Gürcü Valley, lower Miocene, Turkey, bearing a few spots in the abdominal region.

Fig. 5.
Photograph of the isolated group of spots (MNHN 1998.9191 from Agaöz, Gürcü Valley, lower Miocene, Turkey).

We measured preserved tadpoles (kept in the MNHN) of all the recent species listed above and also of Pelobates cultripes (Cuvier, 1829), Pelodytes caucasicus (Boulenger, 1896) and Pelophylax saharicus (Boulenger, 1913) (data not shown) to compare these data with those of Table 1. The results do not allow exclusion of any of these four genera. However, the general shape of the body and of the tail in lateral view suggests that the fossil tadpoles cannot belong to the genus Pelophylax: in the Pelophylax tadpoles the height of the upper tail fin shows its maximum in its proximal part (Nöllert and Nöllert 1992: 114) whereas in the Turkish tadpoles, the maximum height of the upper tail fin is reached more posteriorly. Another frog family should be discussed here, the Palaeobatrachidae Cope, 1865, which were common in Europe during the Tertiary, although not reported from Turkey (Sanchíz 1998). Assignation of the fossil tadpoles to this extinct family can be excluded, as the tadpoles of Palaeobatrachidae exhibit several morphological and anatomical characters which easily distinguish them from those of other families, and especially the Pelobatidae. The most conspicuous are the presence of free ribs, the absence of lateral processes of the parasphenoid, an undivided frontoparietal and the general shape of the body which is reminiscent of the Pipidae Gray, 1825 (Špinar 1972: 38). Furthermore, although not clearly mentioned in the literature, the tadpoles of the Paleobatrachidae should lack keratinised mouthparts, as in all members of the superfamily Pipoidea Gray, 1825.
Wassersug and Wake (1995), reporting about similar tadpoles from a nearby locality, referred them to the genus Pelobates for several reasons that they explained in detail. The evidence in favour of this identification is rather convincing. We observed no character suggesting that our tadpoles would not be conspecific with those described by these authors. However, these authors did not discuss the possibility that these tadpoles belong to the extinct pelobatid genus Eopelobates. Maus and Wuttke (2002) compared osteological features of tadpoles of Pelobates and Eopelobates and found two characters which allow discrimination of the two forms: the shape of the frontoparietal bones, which possess a processus lateralis superior in Pelobates species but not in Eopelobates, and the width/length ratio of the vertebrae II–IV which is between four and five in Pelobates and between five and seven in Eopelobates. The specimen MNHN 1998.9176 in which the frontoparietals are well preserved shows a processus lateralis superior on the right one (the tadpole being slightly in latero-dorsal view, this processus is hidden in the left one). The vertebrae II and III of the same specimen, although their length is not very well outlined, have a width/length ratio slightly below five.
Since all these data do not contradict the hypothesis of Wassersug and Wake (1995), we propose to adopt it for our tadpoles also, pending its possible refutation. Rückert-Ülkümen et al. (2002) reached the same conclusion for similar tadpoles from a nearby locality. Furthermore, as stated above, our sample is composed of tadpoles encompassing a large size range but seemingly of about the same developmental stage. The presence of tadpoles of different sizes in a waterbody seems to be typical for Pelobates fuscus, which is an additional argument for the allocation of the fossil tadpoles to the genus Pelobates (Frank Glaw, personal communication 2009).
Hypotheses about the spots.—Several hypotheses can be put forward to try and explain these spots. Let us first note that these were observed in two distinct localities which are more than 2 km apart (Fig. 2), so that the cause responsible for their appearance cannot be taken as exceptional, e.g., linked to a particular event or to closely related tadpoles (i.e., produced by a single female).
Artefacts.—The dark spots could be non-organic artefacts produced during the fossilisation process, such as gas vesicles originating from decomposition of the intestinal contents of the tadpoles. However, we do not know of any other occurrence of such spots and we are not aware of a natural process that could generate such regular spots. Furthermore, the dark aspect of these spots suggests a protein-rich structure, which does not fit with this hypothesis.
Intestinal contents.—The location of the spots within an area of the tadpole's body (lower posterior part of abdomen) that in normal tadpoles is entirely occupied by the coiled intestine, suggests that the spots could correspond to structures that were part of the intestinal contents. Usually intestinal contents of beaked anuran tadpoles are composed of pieces not exceeding 6 mm in diameter (Heyer 1973: fig. 8), but only few works have been devoted to the detailed study of the nature of tadpoles' intestinal contents. Among the other tadpoles found in Gürcü, intestinal remains are visible in some specimens (MNHN 1998.9176, 9179) as loose filamentous structures, very different from the dark spots here discussed. A striking feature of these dark spots is their regularity in shape, but with a rather high size variability (diameter from 0.76 to 1.6 mm). Such spots could possibly be viewed as a mass of seeds that would have been swallowed and kept unaffected by the whole digestion process. However, the spots appear darker than all vegetal remains present in the slabs, suggesting richer protein content than usually present in vegetal material. Estes et al. (1978) reported on small hollow spherical bodies lateral to the right posterior region of the vertebral column of a Shomronella jordanica Estes, Špinar, and Nevo, 1978 (Pipidae) tadpole in dorsal view. These authors interpreted these structures as part of the intestinal content. Independent of their arguments to reach this conclusion, these structures are quite different from those observed in our samples in that they are clearly hollow in the Shomronella Estes, Špinar, and Nevo, 1978 tadpole whereas the roundish spots discussed here are uniform. Other possibilities for intestinal contents would include either inert structures or bacterial, fungal or algal aggregations developed on intestinal vegetal debris, or eggs.
Internal or external parasites.—We are not aware of any published observation of parasitic infection of tadpoles that would produce a mass of numerous parasitic cysts of regular shape that would correspond to those observed in our two specimens. As for the possible hypothesis of external parasites that would have been fixed to the skin of the belly of the tadpoles, it would not correspond to any known observation in the living fauna. This is quite unlikely given the very thin skin of this part of the tadpole's body, which presumably would not long resist the fixation of external parasites without leading to a tearing of this skin and to the death of the tadpoles.
Disease.—The spots could be thought of as pathological nodules, such as tumours. We do not know of any known case of multiple nodules of similar and regular aspect in tadpoles.
Eggs.—The hypothesis that seems to best fit the observations is that the dark spots correspond to eggs that would have grown in the abdomens of the two tadpoles MNHN 1998.9173–9174. This would explain the large number of spots of similar aspect, with a roundish shape. Eggs would be composed mostly of vitellus, a protein-rich material likely to produce a dark fossil trace. The two groups of eggs associated with tadpoles would still be located in the ovaries, whereas the third group not clearly located within a tadpole might possibly represent a clutch of eggs after its laying by another tadpole. Although less unlikely than the other hypotheses that we have considered above, this hypothesis still raises a number of questions, that will now be considered in more detail.
A review of fossil lower vertebrate eggs.—In his review of fossil vertebrate eggs, Hirsch (1994) did not mention anamniote eggs. However, a few reports of anamniote aquatic vertebrate fossil eggs exist in the literature.
Grande (1984: 101, fig. 2.46) reported the discovery of a specimen of, probably, Diplomystus dentatus Cope, 1877, a clupeomorph teleost, fossilised inside the eggs, from the early Eocene of Wyoming (USA). He provided a photograph of a small fossil teleost curled up inside a light coloured spot. The “eggs” are 12 mm in longest diameter. The fish does not appear to be inside an egg because of its size (about 18 mm) and its degree of ossification (the caudal fin and pectoral girdle are present). Clupeomorph eggs are tiny, never exceeding 2 mm in diameter in extant taxa. We suggest the spot has another origin.
Gall (1991: fig. 2) found “fish eggs” in the Grès à voltzia Formation from the lower Triassic of France. The extremely well preserved fossils show eggs, probably of vertebrates.
Martill (1993) described fossil eggs from the Early Cretaceous Santana Formation of Brazil. He provided SEM micrographs of what undoubtedly are three-dimensional preserved eggs probably produced by an actinopterygian.
Mamay (1994) described Pennsylvanian fossil eggs from New Mexico (USA) of probable chondrostean origin that he considered “the oldest known vertebrate egg masses”.
Clément (1999) provided an explanation of structures sometimes found in the abdominal region of fishes (sarcopterygians as well as actinopterygians). These structures appear as rounded, regular traces. After finding these structures in a fossil coelacanth, he considered the possibility of them being eggs, but he discarded this explanation for several reasons, and favoured a gas origin for the structures. This interpretation explains why traces are often carved in bones.
Finally, three reports of fossil eggs assigned to amphibians are known to us.
Godfrey (1992, 1995) described fossil eggs from the Pennsylvanian of Illinois (USA) that he considered as possible amphibian eggs from now extinct groups.
Mamay et al. (1998) described eggs from the Lower Permian of Texas (USA) that he assigned to the dissophoroid temnospondyl amphibians, also an extinct taxon.
The last reference is the only one that concerns fossil anuran eggs. Špinar (1972) provided photographs of what he thought to be eggs, placed at both sides of the abdominal cavity of an adult anuran from the Tertiary of central Europe. However, both their size and the fact that they appear as carved traces in bones identical to those known in fishes suggest that the same phenomenon implied by Clément (1999), i.e., gas vesicles, was responsible for these traces.
Thus, none of the observations yet published bears a close resemblance with our observations on the Gürcü Valley tadpoles.
Critical study of the egg hypothesis.—First of all, it should be stressed that if the dark spots observed here really represented eggs in ovaries of tadpoles, this would be an extraordinary discovery. In amphibians, this phenomenon is well known in urodelans, but very rare in anurans. Maturation of gonads containing mature gametes have been observed in giant, non metamorphosing specimens of laboratory breeding colonies of Xenopus leavis (Rot-Nikcevic and Wassersug 2004) and in natural populations of the European water frogs of the Pelophylax esculentus complex (Borkin et al. 1982).
The ability for an organism to reproduce while being still in a larval condition (in regards to somatic features) is part of the vast array of developmental phenomena under the general term of aneuchrony (Dubois 1987), i.e., in which chronology of development is modified compared to ancestral forms. This modification may have two different aspects: (1) acceleration or retardation of development relative to ancestors; (2) presence or absence of dissociation between traits (e.g., somatic vs. gonadal) during chronology of development relative to ancestors. Aneuchrony is generally referred to as heterochrony (e.g., Gould 1977; Pierce and Smith 1979; Alberch et al. 1979), but the latter term should better be used to refer to dissociation between traits during the chronology of ontogeny, so that homochronic aneuchrony (without dissociation between traits) can be distinguished from heterochronic aneuchrony (with dissociation between traits) (Dubois 1987).
Paedogenesis (as defined by Dubois 1987) is just one among several possible types of dissociation between traits during ontogeny, in which sexual maturity (mature gonad), with emission of eggs liable to develop, is attained by an organism still showing an overall larval phenotype. This is a rather common phenomenon in urodelan amphibians, but it has never been described in anurans. In the latter group, only a limited number of cases of “partial paedomorphism” (Dubois 1987) have yet been reported, including adult anurans still showing a tail (Olivier 1893), or with a forelimb still emprisoned under skin (Richardson and Barwick 1957; Dubois 1979), or with an intestine still coiled like in tadpoles (Breuil 1981). Similar phenomena (e.g., retention of tail after metamorphosis) were also observed in transgenic tadpoles (Huang et al. 1999; Huang and Brown 2000), or induced by chemical treatment (Elinson et al. 1999), or found in rare athyroid tadpoles appearing spontaneously in laboratory colonies (Rot-Nikcevic and Wassersug 2004). The latter died at the stage when the tail would normally be resorbed.
All these are cases of isolated larval somatic characters retained in an otherwise adult organism, but no cases are known in anurans of the reverse situation, i.e., reproduction in larval organisms. Wassersug (1974, 1975) suggested that this was impossible in anuran tadpoles for anatomical reasons, the abdomen being completely filled up with the intestine, which would leave no space for the development of the gonads. One purely theoretical possibility would be to provide space for the development of the gonads by ceasing to feed, as tadpoles kept without any food tend to show a depletion of the abdomen (before finally dying). The fact that some tadpoles, under special conditions, proved able to develop mature gonads, although without ability to reproduce, raises other questions.
If this phenomenon was possible, why has it never been observed in nature in tadpoles of living anurans species? Such a possibility, if it existed, would have been likely to have been “exploited” in the course of the evolution of anurans, as it has been in that of the urodelans. In the latter group, paedogenesis occurs in some taxa subject to very severe environmental constraints in the aerial stage of their cycle, whereas the aquatic environment is rich and stable. As summarised by Gould (1977), for example, in such conditions, slow growing individuals keep their larval habitat rather than trading it for a hazardous environment. This adaptive response proved so efficient in urodelans that it affected not only some populations and species, but became fixed in more important clades: some families of this order (Proteidae Gray, 1825 and Sirenidae Gray, 1825) are considered to be paedogenetic in origin (Duellman and Trueb 1985; Frost et al. 2006). The fact that not a single case of paedogenesis (heterochrony with possibility of reproduction) is known in anurans suggests that it is unlikely that the Gürcü tadpoles, even if they developed eggs in their abdomen, were able to reproduce. Several more precise questions may be asked in this respect.
Dimension of the spots.—The diameter of the dark roundish spots was estimated to about 1 mm. Such a diameter is smaller than that of the vitellus of mature eggs of the genera Alytes, Pelobates, and Pelodytes laid by adults during reproduction, which are respectively of 2.2–7.0 mm, 1.5–2.5 mm and 1.4– 2.1 mm (van den Elzen 1976; Arnold and Burton 1978; Alcover et al. 1984; Tarkhnishvili 1993). Thus if the tadpoles belonged to one of these genera, the size of the spots would be smaller than that of normal mature eggs of these genera, and it is unlikely that such eggs would be able to develop normally into viable organisms. The dark spots could thus represent immature oocytes. The only case in which the round spots could correspond to mature eggs would be if the studied tadpoles belonged to the genus Pelophylax, as mature eggs of frogs of this group are particularly small, with a vitellus diameter of 0.92– 2.5 mm (Chambers 1908; Arnold and Burton 1978; Berger and Uzzell 1980; Tarkhnishvili 1993; Tsiora and Kyriakopoulou-Sklavounou 2002; Miaud and Muratet 2004). Reasons for exclusion of this genus are discussed above. Furthermore, ovulated eggs in the body of anurans are always densely packed together, which is not the case of the spots under study, which show much space between them in two of our three samples (Fig. 4, 5).
At metamorphosis, most anuran ovaries contain oogonia and primary diplotene oocytes of different sizes (Al-Mukthar and Webb 1971: Xenopus laevis; Lopez 1989: Bombina orientalis (Boulenger, 1890); Ogielska and Wagner 1990: Pelophylax ridibundus; Viertel and Richter 1999. Oocytes at metamorphosis reach 100–130 urn in P. ridibundus (Ogielska and Wagner 1990; Wagner and Ogielska 1990). After metamorphosis, oogenesis and vitellogenesis (Wallace 1978; Wassermann and Smith 1978) and folliculogenesis (Guraya 1978) continue in a species-typical manner until and throughout the adult reproductive life (Echeverría 1988: Rhinella arenarum [Heusel, 1867]; Wagner and Ogielska 1990: P. ridibundus; Van Gansen 1986). Vitellogenesis starts after the first hibernation, whereas the first mature oocytes can be observed just before the third hibernation (Wagner and Ogielska 1990: P. ridibundus).
In conclusion, these data suggest that the spots, if they correspond to female reproductive cells, could not be mature oocytes likely to be fertilised and to develop into tadpoles.
Position of the spots.—The dark spots are situated in a posterior and ventral position in the abdomens of the two tadpoles concerned. This is not the place where ovaries and eggs contained in them would be expected to occur, should ovaries be liable to develop in larval anurans. In adult anurans, ovaries are situated along the vertebral column, often hidden by the kidney, and at any rate in a more anterior and dorsal than posterior and ventral position, the posteroventral part of the abdomen being occupied, in adults like in tadpoles, by the intestine. One possibility would be that the spots represent eggs that were no longer included in the ovary, but had already been ovulated and were about to be laid. Egg-laying in frogs normally requires that the eggs go through the oviduct, where they are given a jelly-coat that will expand in water and protect the vitellus in the external environment. Some of the connections that appear between the spots in the group of spots that was possibly not included in a tadpole could be interpreted as traces of jelly strings, like those that exist in the genera Alytes, Pelobates, and Pelodytes. But this interpretation would not hold for the spots that appear inside the tadpoles, which also show such connections: as long as anuran eggs are included within the female's oviduct, their jelly coat remains very thin, and it only expands with the contact of water outside the female (Lee 1964; Duellman and Trueb 1985: 114).
The anatomical limitations mentioned above, i.e., the lack of space within the abdomen of tadpoles that makes it unlikely that an ovary could expand there while its oocytes grow, also hold for the oviduct. At metamorphosis, the oviduct of frogs is a very thin translucent linear structure, the Müller's canal, which only expands and becomes convoluted at the time of sexual maturity, i.e., much later than metamorphosis (Bhaduri 1953; Bhaduri and Basu 1957; Dubois 1976; Wake 1979; Viertel and Richter 1999). It is difficult to imagine how such a struture, not to mention an ovary full of large eggs, could develop within the abdomen of a tadpole without leading to a change of shape of this abdomen.
Stage of tadpoles.—The two tadpoles showing dark spots in their abdomens are approximately at Gosner stages 36–38. All the 17 other tadpoles, without spots, are at identical developmental stages, but some of them have a larger size (Table 1), so that it cannot be argued that eggs are present in the oldest tadpoles. With a sex-ratio of 1 male / 1 female, the expected number of female tadpoles would be nine or ten, and the difference between this number and the observed number of two is highly significant (χ2 = 9.1; 0.001 < P < 0.005). Thus, if eggs are indeed present in some tadpoles of this population, this would not appear as a regular phenomenon, as it does not seem to affect all female tadpoles. A related question is the absence of young developmental stages among tadpoles: if some tadpoles in this population were laying eggs at the time of fossilisation, and if these eggs had been fertilised, one would expect some of these eggs to have given birth to young, tiny tadpoles; but of course the latter may have escaped fossilisation easier than larger tadpoles.
Other considerations.—The egg hypothesis can be further complicated by other hypotheses, which appear even more difficult to test. It could be imagined that, if tadpoles can develop eggs within their abdomen while being devoid of oviducts for their evacuation, such tadpoles could lay their eggs in the external environment through ventral tearing or “explosion”, as occurs in some invertebrates. In some lower vertebrates, the Petromyzontidae Bonaparte, 1832 and the Myxinidae Bonaparte, 1832 (Agnathes), the oviducts are absent: the mature eggs pass through abdominal pores, which connect the coelomic cavity to the cloaca and are expelled outside by a rupture of the cloaca (Gérard 1958).
As another possibility, the eggs could have been laid by a normal adult amphibian, and two tadpoles would have swallowed such eggs but kept them undigested despite their progression through their digestive ducts, which seems quite unlikely. A single case of adults swallowing eggs but not digesting them is known in anurans, in the now extinct genus Rheobatrachus (Tyler 1983), where “gastric brooding” used to occur, but nothing similar is known in tadpoles. Furthermore, tadpoles that feed on eggs by shallowing them as a whole usually exhibit morphological specialisations (see Suggestions section below).
The question may be raised whether the eggs, if eggs were involved, were indeed inside the tadpoles before fossilisation. Clutches of eggs of amphibians are special environments that often attract organisms as a shelter, providing a warmer place (according to the conditions, frog egg masses may be several degrees warmer than the surrounding water) or a source of food: often aquatic insects, fishes or amphibians (adult newts or frogs) are found in clutches of frog eggs, especially in cold waters (AD, personal observations). The two tadpoles MNHN 1998.9173–9174 could have been fossilised within egg masses and the eggs become superimposed on their abdomens during fossilisation. All these hypotheses are quite unlikely, but they are mentioned here in order to point to the uncertainties that remain regarding the interpretation of these observations.
Finally, let us stress that if eggs were laid by paedogenetic tadpoles, this does not mean that these eggs could develop: for this, they would need to be fertilised, which would require either complete paedogenesis in male tadpoles of the same species, or fertilisation by adult males—except if, beside being paedogenetic, these extraordinary tadpoles were also parthenogenetic!
European anurans of the genera discussed above all have a reproductive mode involving an amplexus in which the male grasps the female either above the hindlimbs or behind the forelimbs. Such an amplexus is impossible in tadpoles which are either devoid of free forelimbs (as long as they are covered by the operculum) or with forelimbs too short for grasping the large, ovoid body of the female tadpole. In order to ensure that sperm produced by a male tadpole could fertilise the eggs produced by a female tadpole, a completely new behaviour would need to be established, as well as anatomical structures allowing ejection of the gametes into the aquatic habitat. For paedogenesis to be possible in anuran tadpoles, maturation of gonads is not sufficient, but morphological and behavioural changes would be necessary. Evidence is missing that such major changes ever occurred in anurans. This point is certainly the strongest argument against the possibility of efficient paedogenesis in anurans.
Suggestions for future research.—Although very common organisms over most of our planet, tadpoles have still attracted very little attention. The first book dealing exclusively with these very peculiar and exciting organisms was published only ten years ago (McDiarmid and Altig 1999). As acknowledged by this book, still a lot has to be learned about them. The present observations and the hypotheses they suggest lead us to propose some questions that could be tested in future studies of tadpoles of living amphibians.
(i) Could there be some variation in the development stage reached by the gonad and the oviduct in tadpoles, according to the taxon and the conditions? If in some cases the ovary and oocytes can show some growth, where are they situated in the abdominal cavity? Could this account for the observation reported here?
(ii) Would it be possible to act upon the gonad and oviduct development of anuran tadpoles by chemical factors, e.g., those known to facilitate artifical paedogenesis in urodelans? It is possible to stop the development of anuran tadpoles with aminothiazol, thiouracile or thiourea, or simply by a thyroidectomy, while allowing the growth of gonads (Delsol 1952: Discoglossus pictus Otth, 1837). Similarly, the ethinyltestosterone (pregneninolone) given in aqueous solution to tadpoles exerts a strong inhibiting action on the thyroid, blocking the somatic larval development. However, the masculinising action of this hormone in strong doses allows the development, during larval life, of immature testes (without spermatozoids) and of associated structures (Wolffian ducts and seminal vesicles) (Gallien 1949: Rana temporaria Linnaeus, 1758).
In thyroidless larvae of Lithobates sylvaticus (LeConte, 1825) (formerly Rana sylvatica, Ranidae; see Frost et al. 2006) kept through the second season after the controls have metamorphosed, the ovaries became large and large oocytes developed, although maturation was not observed and oviducts did not develop (the animals were not sexually mature). The testes became mature and produced spermatozoa which escaped into the kidneys (Hoskins and Hoskins 1919).
In similar experiments using thyroidectomy in Lithobates pipiens (Schreber, 1782) (formerly Rana pipiens, Ranidae), Allen (1917, 1918) demonstrated that spermatogenesis proceeds normally and sexual maturity is attained in thyroidless male tadpoles that have retained larval characteristics long after the time of metamorphosis. The ovaries of the female thyroidless tadpoles increased in size with time until they had reached a volume well in excess of that of the young normal frogs killed shortly after metamorphosis. The oocytes showed a diameter double of that in the recently metamorphosed frogs. In these experiments, artificial paedogenesis was definitely produced: sexual maturity was reached in the male and it is probable that it would have been reached in the course of time in the female tadpoles.
Some giant tadpoles from natural populations in Latvia showed “well-developed eggs in ovaries” and “testes normally developed” but no hindlimbs and a strongly pigmented skin (Borkin et al. 1982).
Finally, in a recent paper, Rot-Nikcevic and Wassersug (2004) reported about thyroidless tadpoles of Xenopus laevis occurring spontaneously in laboratory breeding colonies. These giant tadpoles stopped their somatic development and never metamorphosed. They showed an advanced gonadal development. The testes contained sperm, and the ovaries mature eggs with yolk. However, these tadpoles developed excessive musculature (especially the axial muscle mass of the tail) and had a scoliosis which deformed the back in dorsal view. This is obviously not what we observed in the fossil tadpoles described here.
The absence of thyroid hormones, while the hypophysis is still secreting prolactin and growth hormones, stops the somatic development although the development of the germ-line continues. The dissociation of the development of germen and soma allows the gonads to have time to reach maturity as the tadpole's size increases (Rot-Nikcevic and Wassersug 2004). Therefore, the largest tadpoles are usually the oldest and show the most advanced gonadal maturity. As the fossil tadpoles containing the black spots presented here were not the largest ones, this suggests that they are probably not thyroidless tadpoles.
(iii) Can some tadpoles, in some conditions, swallow whole amphibian eggs, or vegetal seeds, and keep them undigested through the intestinal duct? Could such a phenomenon explain the observations reported about here?
Tadpoles feeding from unfertilised eggs from their own mother are known in six anuran families: Dendrobatidae Cope, 1865 (Oophaga pumilio [Schmidt, 1857]: Weygoldt 1980; Crump 1983), Hylidae Rafinesque, 1815 (Noble 1929; Dunn 1937; Taylor 1954; Crump 1983; Lannoo et al. 1987; Jungfer and Schiesari 1995), Leptodactylidae Werner, 1896 (Leptodactylus pentadactylus group species: Muedeking and Heyer 1976; Hero and Galatti 1990; Magnusson and Hero 1991; Silva et al. 2005), Mantellidae Laurent, 1946 (Mantella laevigata Methuen and Hewitt, 1913: Glaw et al. 2000; Heying 2001), Microhylidae Günther, 1858 (Hoplophryne Barbour and Loveridge, 1928: Noble 1929; Crump 1983) and Rhacophoridae Hoffman, 1932 (“Philautus” cf. carinensis: Crump 1983; Kurixalus eiffingeri [Boettger, 1895]: Ueda 1986; Kam et al. 1996). However, the tadpoles of Oophaga pumilio feed on the eggs by biting a hole through the jelly capsule and sucking up the contents (Weygoldt 1980), so that the eggs are not wholly engulfed and would not be found complete in the gut. Similarly, Mantella laevigata eats the conspecific eggs and so does not swallow them (Glaw et al. 2000; Heying 2001), and only fragments of eggs were found in the guts of Hoplophryne tadpoles (Noble 1929). Tadpoles of the Leptodactylus pentadactylus species group also feed on heterospecific eggs. Leptodactylus labyrinthicus (Spix, 1824) tadpoles are reported to swallow entire eggs of Dendropsophus minutus (Peters, 1872), Physalaemus cf. fuscomaculatus (Steindachner, 1864). and P. cuvieri Fitzinger, 1826 (Silva et al. 2005). In Osteopilus brunneus (Gosse, 1851), from 5 to 184 whole eggs can be found in the stomach of the tadpoles, and a few intact eggs even in the intestine (Lannoo et al. 1987). In Isthmohyla zeteki (Gaige, 1929), at least 31 conspecific eggs were found in a stomach of a 9 mm head-body length tadpole (Dunn 1937). In the stomach of an unidentified rhacophorid species (“Philautus” cf. carinensis), 13 frog eggs with a mean diameter of 1.43 mm were found (Wassersug et al. 1981). No data about the number and size of the engulfed eggs were provided for the case of Anotheca spinosa (Steindachner, 1864) tadpoles (Taylor 1954), but according to the drawings, whole eggs are clearly visible through the body wall and localised posteroventrally in the abdominal cavity, reminiscent of the structures observed in this study. Oophagous tadpoles usually exhibit morphological features which appear adaptive relative to this feeding habit, such as a few keratodonts arranged in a few rows, a powerful head musculature, a short gut and an enlarged stomach. All the cases of cannibalism just listed concern tadpoles which develop in tree holes or in bromeliads, small habitats almost completely devoid of nutritive matter. This is a different situation from that of the Turkish tadpoles here described, which apparently lived in large lakes or swamps where organic material was presumably abundant.
However, a few cases are known of opportunistic cannibalism in tadpoles in temporary puddles. This was reported in Isthmohyla pseudopuma (Günther, 1901) by Crump (1983). Tadpoles preying on clutches of other frog species have also been reported (Heusser 1970, 2000; Mutz 2005), but, although these papers did not provide details, it is very likely that the eggs are rasped and therefore destroyed before being swallowed.
Conclusion
Two of the 19 fossil tadpoles from the Miocene of Turkey here reported show unusual groups of regular roundish black spots in the abdominal region. Several possible hypotheses regarding the origin of these spots are discussed. None is fully satisfactory. In the improbable case where these spots would correspond to eggs, this would be the first reported case of paedogenesis in anurans, a phenomenon which has been so far considered impossible mostly for anatomical reasons (i.e., absence of space in the abdominal cavity). In the future, it would be interesting to explore this possibility, both experimentally and by broad anatomical survey of many tadpoles, both of living species and in fossil material.
Acknowledgements
René Zaragüeta (UPMC, Paris, France) prepared the drawing of Fig. 3B and provided bibliographic references. Gaël Clément (MNHN, Paris, France), Martine Desoutter-Meniger (MNHN, Paris, France), Jean-Claude Gall (Université Louis Pasteur, Strasbourg, France), Frank Glaw (ZSM, München, Germany), Philippe Janvier (MNHN, Paris, France), Julien Marmayou (MNHN, Paris, France), Andrew R. Milner (NHM, London, UK), André Nel (MNHN, Paris, France), Annemarie Ohler (MNHN, Paris, France) and Jean-Claude Rage (MNHN, Paris, France) provided bibliographic help, translations, information on some of the slabs from Beşkonak, help for the figures and/or useful suggestions on the manuscript.