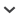The giant cardon cactus, Pachycereus pringlei (S. Watson) Britton & Rose (Cactaceae), is a columnar cactus that is widely distributed in the Sonoran Desert in Mexico, including the Baja California peninsula, all islands in the Gulf of California, and the coastal region of Sonora State (Turner et al., 1995). This species offers an attractive model for evolutionary studies. Its wide distribution is characterized by a heterogeneous array of environmental conditions (Garcillán et al., 2010), as well as a geographic variation in the distribution of its major pollinator, the lesser long-nosed bat Leptonycteris curasoae (Fleming et al., 1998). Heterogeneity in biotic and abiotic factors can result in differential selective pressures across populations, promoting genetic differentiation and local adaptation. For P. pringlei, a latitudinal pattern of abundance, height, and basal diameter in the Baja California peninsula is associated with annual and summer rainfall, as well as winter temperature (Medel-Narváez et al., 2006). This species also exhibits an unusual geographical variation of breeding systems among Cactaceae, including hermaphroditism (Medel-Narváez, 2008), gynodioecy (female and hermaphrodites), and trioecy (males, females, and hermaphrodites). This spatial segregation of sexes is partly associated with variations in abundance of its major pollinator (Fleming et al., 1998).
The Baja California peninsula had a dynamic geologic history, including the formation of the Gulf of California approximately 5.5 mya (Lonsdale, 1989) and major climatic changes during the Pleistocene (Van Devender, 1990) that have had a profound effect on the distribution and genetic structure of other columnar cacti with similar ranges of distribution, such as Lophocereus schottii (Engelm.) Britton & Rose (Nason et al., 2002) and Stenocereus gummosus (Engelm.) A. C. Gibson & K. E. Horak (Clark-Tapia and Molina-Freaner, 2003). Currently, there is one molecular study based on low polymorphic markers (allozymes) that suggests no genetic differentiation within the range of P. pringlei (Fleming et al., 1998). In this study, we used the latest 454 GS-FLX titanium sequencing platform to locate polymorphic microsatellite loci in P. pringlei and propose that these molecular markers be used in genetic studies.
METHODS AND RESULTS
Genomic DNA was extracted from fresh cortex tissue of one P. pringlei cactus growing near La Paz, Baja California Sur, Mexico (23.985°N, 110.187°W), using a procedure with cetyltrimethylammonium bromide (CTAB) modified for mucilaginous cactus tissues (de la Cruz et al., 1997). A voucher specimen (JLLL11726 [HCIB28440]) was deposited in the Herbarium HCIB at Centro de Investigaciones Biológicas del Noroeste (CIBNOR). Approximately 5 µg of genomic DNA was treated with RNAse and submitted to the Genome Sequencing Center at the University of California, Santa Cruz, where the sample was sequenced using 454 pyrosequencing with the GS-FLX titanium platform (Roche Diagnostics, Basel, Switzerland), along with seven other species. The alignments were run according to the barcode plate strategy, as outlined in the GS-FLX Shotgun DNA Library Preparation Method Manual (supplied by Roche Diagnostics, Roche Applied Science, Mannheim, Germany).
Table 1.
Characteristics of 10 microsatellite loci developed for the cardon cactus, Pachycereus pringlei.

All 454 reads were first converted from flowgrams to FASTA and FASTA-qual files without 454 adapters, using the sff_extract software ( http://bioinf.comav.upv.es/sff_extract/index.html). We found 398,286 sequence reads with an average read length of 274 bp from two independent runs. MSATCOMMANDER software (Faircloth, 2008) was used to identify perfect microsatellites containing at least five repeat units for dinucleotides and four for tri-, tetra-, penta-, and hexanuclotides. Perfect microsatellite motifs were found in 10,685 (2.6%) reads, of which dinucleotide repeats were most abundant (62%), followed by trinucleotide repeats (31%), and tetranucleotide repeats (6%). Using Primer3 software (Rozen and Skaletsky, 2000) embedded in the QDD software (Meglécz et al., 2010), primer design was possible in 282 reads, with the following parameters: (1) melting temperatures 50–70°C, (2) PCR product between 90 and 320 bp, (3) GC content >40%, (4) primer length between 17 and 27 nucleotides, (5) primer self-complementarities and complement between them fulfill the quality criteria used as default parameters in the Primer3 software.
Table 2.
Genetic diversity of the 10 microsatellite loci developed from the cardon cactus, Pachycereus pringlei.a

Favoring larger motif (repetition numbers) and lower melting temperature differences between pairs of primers, we selected 38 loci for PCR amplification and a banding pattern of 10 cacti samples collected at El Comitán near La Paz (23.985°N, 110.187°W). Total DNA was extracted from cortex tissue, using the modified CTAB procedure mentioned earlier. PCR reactions were performed in 10-µL volumes using a thermocycler (ICycler, Bio-Rad Laboratories, Hercules, California, USA). Each reaction contained 50 ng of DNA, 0.2 mM of each dNTP (no. 18427-088, Invitrogen, Carlsbad, California, USA), 0.4 µM of each primer (Applied Biosystems, Carlsbad, California, USA), 1× PCR buffer (20 mM Tris-HCl, 50 mM KCl [pH 8.4]), 1–2.5 mM MgCl2, 0.4 U Taq DNA polymerase (no. 11615-010, Invitrogen), and 0.1 mg mL−1 of bovine serum albumin (BSA; no. B90015, New England Biolabs, Ipswich, Massachusetts, USA). Cycling conditions included initial denaturation at 94°C for 5 min, 30 cycles at 94°C for 1 min each, 45 s at the locus-specific annealing temperature (see Table 1 and Appendix S1 (APPS-D-13-00066_AppS1.docx)), extension at 72°C for 1 min, and a final extension at 72°C for 25 min. PCR products were visualized by silver staining in 6% Polyacrylamide (Bio-Rad Laboratories), 7.5 M urea, and a 10-bp ladder (Invitrogen). Results of PCR amplification of the 38 pairs of primers are given in Appendix S1 (APPS-D-13-00066_AppS1.docx).
Ten of the 38 primer pairs yielded distinct bands in the expected size range; consistent amplification patterns and fewer stutter bands were selected for characterization in 40 individuals that were sampled at El Comitán and 40 that were sampled at Punta Prieta, Ensenada, Baja California (29.063°N, 114.155°W). Duplicate samples of each individual were stored in 96% ethanol at the Laboratorio de Genética para la Conservación at CIBNOR. PCR reactions were performed, as described above, using optimized primer-specific annealing temperatures (Table 1), magnesium chloride, and forward-labeled primers (5′ end-labeled with FAM, NED, PET, and VIC; Applied Biosystems). Individuals were genotyped by assessing allele size on a genetic analyzer (model 310, Applied Biosystems), using the GeneScan 600 LIZ Size Standard (Applied Biosystems) and GeneMapper 4.1 software (Applied Biosystems).
Locus designation, primer sequences, repeat motifs, annealing temperatures, PCR product sizes, and GenBank accession numbers are listed in Table 1. All 10 loci were polymorphic, exhibiting two to 10 alleles per locus (mean A = 5.5) among cacti from El Comitán, and two to seven alleles per locus (mean A = 3.7) among cacti from Punta Prieta. Alleles per individual plant ranged from one to four in both populations, suggesting a polyploidal genome (Table 2).
Observed heterozygosity was estimated in Microsoft Excel (Microsoft Corporation, Redmond, Washington, USA). Expected heterozygosity was estimated in ATETRA software for tetraploid species that account for all possible combinations of allele copy numbers in populations with partial heterozygotes (Van Puyvelde et al., 2010). Genetic diversity in terms of heterozygosity varied from low to high across loci in both populations. Observed heterozygosity ranged from 0.13 to 0.78 (mean Ho = 0.59) and expected heterozygosity ranged from 0.37 to 0.77 (mean He = 0.57) at El Comitán. At Punta Prieta, observed heterozygosity ranged from 0.03 to 0.85 (mean Ho = 0.41) and expected heterozygosity ranged from 0.03 to 0.72 (mean He = 0.40) (Table 2). For this study, we assumed tetraploidy in both populations because we observed as many as four alleles per locus in individual plants. Our results are also consistent with a tetrasomic pattern of inheritance previously suggested in P. pringlei, based on an allozymatic study (Murawski et al., 1994) and a chromosome counting study (Pinkava et al., 1973).
CONCLUSIONS
The 10 microsatellites are the first to be developed for the cardon cactus, P. pringlei, and the genus Pachycereus. These polymorphic loci will be useful in studies of genetic diversity and genetic population differentiation and provide valuable information to understand the importance of biotic and abiotic factors involved in geographic variation in the genetic pool of this widespread cactus of the Sonoran Desert.
LITERATURE CITED
Notes
[1] The authors thank Raymundo Domínguez-Cadena and Alfonso Medel-Narváez for their field assistance, Carlos Garza of the National Oceanic and Atmospheric Administration (NOAA) for providing sequencing service, and Ira Fogel of Centro de Investigaciones Biológicas del Noroeste (CIBNOR) for providing editorial assistance. The Secretaría del Medio Ambiente y Recursos Naturales (SEMARNAT) issued collection permits (SGPA/DGVS/07004/11). Funding was provided by the Consejo Nacional de Ciencia y Tecnología (CONACYT; grant CB-2008-01-106925). C.G.F. is a recipient of a CONACYT student fellowship (165852).