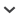Nicotinic acetylcholine receptors (nAChR) play major roles in the fast synapse cholinergic transmission in the insect central nervous system, and they are recognized as potential target sites for insecticides. In this study, the full-length cDNA sequence of the nAChR alpha6 subunit (Bdorα6) was isolated and characterized from the oriental fruit fly, Bactrocera dorsalis, using the Rapid Amplification of cDNA Ends (RACE) technique. The Bdorα6 cDNA was 2020 bp long, containing an open reading frame of 1467 bp, which encodes 488 amino acid residues with a predicated molecular mass of 55.6 kDa. Homology analysis indicated that Bdorα6 has typical features of a nAChR α subunit. Phylogenetic analysis with other insect nAChR subunits suggested that Bdorα6 was most closely related to Dα6 of Drosophila melanogaster. The developmental changes and tissue-specificity of the relative expression levels of Bdorα6 were investigated using quantitative real-time PCR. The highest expression level was observed in the first instar larva and the lowest was in the pupa, and there was a descending expression level during the developmental period from the first instar larva to the pupa. The relative expression levels were 23-, 39-, 6-, 2- and 27-fold higher in the egg, first, second and third instar larvae and adult than in the pupa, respectively. Furthermore, Bdorα6 was expressed in the head and thorax at 109- and 15-fold higher levels than in the abdomen. The results provided some important baseline data for further molecular studies in Bactrocera dorsalis.
Nicotinic acetylcholine receptors (nAChR) play major roles in the fast synapse cholinergic transmission in insect central nervous system (Gundelfinger & Schulz 2000). The nAChRs belong to the cys-loop superfamily of ligand-gated ion channels, which also includes γ-aminobutyric acid (GABA)-gated Cl- channels, glycine receptors, glutamate-gated Cl- channels, and 5-hydroxytryptamine type 3 (5-HT3) receptors (Lester et al. 2004). The nAChRs consist of 5 homologous subunits, typically 2 α subunits and 3 non-α subunits, but receptors consisting of only α subunits are also known (Couturier et al. 1990; Marshall et al. 1990). The α subunits are characterized by the presence of 2 adjacent cysteine residues in loop C, while the non-α subunits lack this cysteine doublet (Gotti & Clementi 2004; Sattelle et al. 2005). Each subunit has an N-terminal extracellular domain that includes a Cys-loop and acetylcholine (ACh) binding site, which is located at the interface of 2 subunits and is formed by 6 loops (loops A–F) with each of the adjacent subunits contributing 3 loops (Grutter & Changeux 2001). Each subunit also contains 4 transmembrane regions (TM1–4), of which TM2 donates most of the residues that line the ion channel, and the intracellular linker between TM3 and TM4, which is responsible for receptor desensitization and intracellular trafficking (Corriger et al. 2000; Karlin 2002).
Based on completed genome sequences, cloning of nAChR cDNAs has been performed for 2 dipteran species, Drosophila melanogaster (7 α and 3 β) and Anopheles gambiae (9 α and 1 β), possessing 10 nAChR subunit genes (Littleton & Ganetzky 2000; Jones et al. 2005), for the hymenopteran species, Apis mellifera (9 α and 2 β), possessing 11 nAChR subunit genes (Jones et al. 2006), and for the coleopteran species, Tribolium castaneum (11 α and 1 β), and Lepidopteran species, Bombyx mori (9 α and 3 β), both possessing 12 nAChR subunit genes (Jones & Sattelle 2007; Shao et al. 2007). In addition, many other insect nAChR subunit genes have also been identified such as Manduca sexta, Myzus persicae, Schistocerca gregaria (Tomizawa & Casida 2001), Ctenocephalides felis (Bass et al. 2006), Sitobion avenae (Qiu & Han 2007), Liposcelis bostrychophila (Tang et al. 2009), and Apis cerana cerana (Yu et al. 2011). The insect nAChRs have been recognized as potential target sites for insecticides such as neonicotinoid insecticides and spinosad (Tomizawa et al. 1999; Salgado & Sparks 2005). For instance, A Y151S mutation was identified in 2 nAChR subunits (Nlα1 and Nlα3) from neonicotinoid imidacloprid-resistant of Nilaparvata lugens and was strongly correlated with a substantial reduction in specific imidacloprid binding (Liu et al. 2005). In D. melanogaster, mutations in Dα1 or Dβ2 nAChR subunits can confer resistance to neonicontinoids, and a knockout of the Dα6 subunit of the nAChR confers high level of resistance to spinosad (Perry et al. 2007; Perry et al. 2008).
The oriental fruit fly, Bactrocera dorsalis (Hendel), is one of the most destructive pests with a cosmopolitan distribution in tropical and subtropical regions. The oriental fruit fly is a polyphagous pest that damages more than 250 plant species including numerous fruits and vegetables (Clarke et al. 2005; Chen & Ye 2007). Measures taken to control the pest mostly involve using chemical insecticides. In recent years, this pest has developed high level resistance to organophosphates, pyrethroids, and avermectin (Hsu et al. 2004; Zhang et al. 2007). Resistance of S. dorsalis to spinosad has been developed in laboratory selection and cross-resistance experiments (Hsu & Feng 2006). Therefore, it is expected that the resistance to spinosad may also increase rapidly in orchards globally.
In order to develop insecticide resistance monitoring tools and understand the mechanism of insecticide resistance targeting nAChR in B. dorsalis, we report the cloning and characterization of the nicotinic acetylcholine receptor alpha6 cDNA and the expression patterns at different developmental stages and in different tagmata of adult flies using quantitative real-time PCR.
MATERIALS AND METHODS
Insects
The oriental fruit fly, Bactrocera dorsalis, was originally collected from Fujian province, People's Republic of China. The adults were reared on an artificial diet consisting of water, yeast powder, honey, and Vitamin C (500:15:15:1) in glass cages. Every 2 days, a banana was put into the cage to collect eggs. After hatching, larvae were reared on banana in plastic basins with sand until pupation. The colonies were kept in a temperature controlled room at 27 ∓ 1 °C, 70 ∓ 5 %RH at 14:10 h L:D throughout development (Shen et al. 2010).
RNA Isolation and cDNA Preparation
Total RNA was isolated from the heads of adult flies (15 mg) using an RNA Isolation Kit (Watson Biotechnologies, Shanghai, China) following the manufacturer's instructions. The total RNA was treated with DNase (TaKaRa, Dalian, China) and dissolved in 40 µL DEPC-treated H2O and stored at -80 °C until use. The first-strand cDNA was synthesized from 2 µg of DNase-treated RNA by RevertAid™ First Strand cDNA Synthesis Kit (Fermentas, Vilnius, Lithuania) with oligo (dT)18 primers according to the manufacturer's instructions. The reaction mixture was stored at -20 °C for future use.
Amplification of cDNA Fragments
Degenerate primers, Bd1, 5′-ATGAARTTYGGNWSNTGGACNTAYGA-3′ and Bd2, 5′-GCNACCATRAACATDATRCARTTRAA-3′, were designed based on the conserved motifs of nicotinic acetylcholine receptor family to amplify the cDNA fragments (Gao et al. 2007b). Degenerate PCR was conducted using rTaq™ polymerase (TaKaRa, Dalian, China) in 25 µL, including 1 µL cDNA templates, 1 µL each primer (10 µM), 2 µL dNTP (2.5 mM), and 2.5 µL 10× PCR buffer (Mg2+ plus). Degenerate PCR amplifications were performed under the following conditions: initial denaturation at 95 °C for 3 min, followed by 34 cycles of denaturation at 95 °C for 30 sec, annealing at 55 °C for 30 sec, and extension at 72 °C for 1 min, with a final extension at 72 °C for 10 min. The PCR products were then separated by 1.5% agarose gel electrophoresis and stained with ethidium bromide (EB). A band of 428 bp cDNA was excised and purified using the Gel Extraction Mini Kit (Watson Biotechnologies, Shanghai).
The purified fragments were cloned into pGEM®-T Easy vector (Promega, Madison, Wisconsin). The ligation reactions were used for transformations with DH5α competent cells (Transgen, Beijing), the DNA inserts of the recombinant clones were identified by PCR with the primers used before, and further confirmed by sequencing in both directions with an ABI-PRISM 3730 sequencer (Invitrogen Life Technologies, Shanghai, China).
Rapid Amplification of cDNA Ends (RACE) and Cloning of the Open Reading Frame (ORF) of Bdorα6
The full-length cDNA of Bdorα6 was obtained by 5′/3′ RACE techniques using a SMARTer™ RACE cDNA Amplification Kit (Clontech, California) according to the manufacturer's instructions. The 3′-RACE-ready cDNA was synthesized from 2 µg of total RNA at 42 °C for 1.5 h with SMARTScribe™ Reverse Transcriptase and the 3′-CDS primer, while the 5′-RACE-ready cDNA was synthesized with SMARTScribe™ Reverse Transcriptase, the 5′-CDS primer, and SMARTer IIA oligo.
Based on the 428 bp cDNA sequence, 4 gene specific primers were designed to amplify the full-length cDNA: 2 forward primers (in the first round reaction, GSP1: 5′-ACAAATGGCGAGTGGTATCTGC-3′ and in the second round reaction, GSP2: 5′-CTTCTATCGCTCACGGTGTTTCTC-3) for 5′-RACE PCR and 2 reverse primers (in the first round reaction, GSP3: 5′-GAGAAACACCGTGAGCGATAGAAG-3′ and in the second round reaction, GSP4: 5′-CGCCGTCGGATTTGTATCGTGAA-3′) for 3′-RACE PCR. Utilizing the gene specific primers, universal primer (UPM, provided by the Clontech kit) and nested universal primer (NUP, provided by the Clontech kit), the 3′-and 5′-RACE were conducted with PrimeSTAR™ HS DNA polymerase (TaKaRa, Dalian, China). Thermal cycling conditions were: pre-denaturation 3 min at 94 °C, 30 cycles of 94 °C for 30 sec, 60 °C -65 °C (according to the annealing temperature of the primer) for 30 sec and 72 °C for 1–1.5 min (according to the size of expected fragment) with a final extension of 10 min at 72 °C. The resulting PCR products of 5′-RACE and 3′-RACE were cloned into pGEM®-T Easy vector (Promega, Madison, Wisconsin) and sequenced as described above.
After the 5′-ends and 3′-RACE sequences were obtained, the contigs were assembled to produce putative full-length sequences by using DNAMAN 5.2.2 (Lynnon BioSoft, Quebec). To verify the full-length cDNA, the primers BdF, 5′-ACATGGACCCGTCGCTGCTAGTC-3′ and BdR, 5′-CATTATCGTGCAATAAGCCTAAA-3′, were designed based on the sequences obtained from 5′-untranslated region (UTR) and 3′-UTR of the gene and used to amplify the ORF of Bdorα6. The PCR products were purified, cloned, and sequenced as described above.
Sequence Analysis
A search for similar sequences was performed using BlastP in the non-redundant protein sequences (nr) database of the NCBI website ( http://www.ncbi.nlm.nih.gov/). Multiple sequence alignment was performed with DNAMAN 5.2.2 (Lynnon BioSoft, Quebec). The signal peptide was predicated by SignalP 3.0 (Bendtsen et al. 2004). Phosphorylation sites and N-linked glycosylation sites were identified by the PROSITE database (Falquet et al. 2002). Transmembrane domains were predicted by the TMHMM program ( http://www.cbs.dtu.dk/services/TMHMM/). The phylogenetic tree construction by MEGA5.04 (Tamura et al. 2011) used the neighbor-joining method. Bootstrap values were calculated on 1000 replications.
Quantitative Real-Time PCR of Bdorα6
The expression levels of Bdorα6 mRNA relative to expression of α-tubulin at different developmental stages and in different tagmata of B. dorsalis were examined using quantitative realtime PCR. Total RNA was isolated from whole bodies at different developmental stages (including egg, first, second and third instar larvae, pupa, and adult) or from heads, thoraces, and abdomens of adult flies, using RNeasy Plus Mini Kit (with gDNA Elimator spin columns, Qiagen, Valencia, California). First strand cDNA was synthesized in a 10 µL reaction mixture using random hexamers by PrimeScript® RT reagent Kit (TaKaRa, Dalian, China). The quantitative realtime PCR was carried out by Mx3000P thermal cycler (Stratagene, La Jolla, California). The PCR amplifications were performed in 25 µL reactions systems containing 2 µL of template cDNA, 12.5 µL iQ™ SYBR® Green Supermix (BIO-RAD, Hercules, CA, USA) and 0.2 mM each of the primers 5′-ATTCGATGGCACGTATCACA-3′ (forward) and 5′-GGGCACCAAGTTAGTCTGGA-3′ (reverse) for Bdorα6, and 5′-CGCATTCATGGTTGATAACG-3′ (forward) and 5′-GGGCACCAAGTTAGTCTGGA-3′ (reverse) for α-tubulin. Amplification conditions were as follows: initial denaturation at 95 °C for 2 min, followed by 40 cycles of denaturation at 95 °C for 15 sec, 60 °C for 30 sec and 72 °C for 30 sec. After reaction, a melting curve analysis from 60 °C to 95 °C was applied to all reactions to ensure consistency and specificity of the amplified product. The real-time PCR analysis was repeated 3 times in independent experiments. The data of different developmental stages and tagmata were normalized according to the expression level of α-tubulin gene (GenBank accession number: GU269902). When we tested the mRNA expression during the different life stages, the pupal stage served as the calibrator sample whose mRNA expression ratio would always equal 1. In addition, when testing mRNA expression in different tagmata, the abdomen served as the calibrator sample.
Statistics Analysis
The PCR efficiencies of Bdorα6 and α-tubulin were calculated by the Mxpro-Mx3000P version 4.01 (Stratagene, La Jolla, California). Data from the quantitative real-time PCR for both the developmental expression and different tagmata expression profiles were subjected to LSD test in analysis of variance (ANOVA) (SPSS 12.0 for Windows).
RESULTS
Cloning and Characterization of Bdorα6
Degenerate primers Bd1 and Bd2 were used to amplify a 428 bp fragment from a cDNA preparation of B. dorsalis. Based on the sequence of the cDNA fragment, we obtained the full-length cDNA through the 5′/3′ RACE technique. It was the closest similarity to other known insect α6-type nAChR subunits at amino acid levels; the gene was named as Bdorα6. The cDNA sequence and deduced amino acid sequence of the Bdorα6 is shown in Fig. 1. The complete cDNA of the Bdorα6 (GenBank accession number: JF974072) consists of 2020 nucleotides with an ORF of 1464 nucleotides encoding 488 amino acids. The cDNA includes a 5′-UTR located 206 bp upstream of the start codon (ATG) and 3′UTR of 350 nucleotides that ended in a poly (A) tail. A possible consensus signal sequence for polyadenylation (AATAAA) is located 27 bp upstream of the poly (A) tail. The theoretical molecular mass of Bdorα6 based on the deduced amino acid sequence was calculated to be 55.6 kDa, with an isoelectric point of 5.41.
We performed a signal peptide scan using the SignalP 3.0 program ( http://www.cbs.dtu.dk/services/SignalP) finding that the protein possesses a predicted 21-residue long signal peptide. The protein has 2 potential N-linked glycosylation sites (N-X-S/T) at positions 44 and 88 in the N-terminal extracellular domain. There are 15 potential phosphorylation sites for protein kinase C, casein kinase II, tyrosine kinase and cAMP- and cGMP- dependent protein kinase, 7 of which are located in the N-terminal extracellular domain, 1 located in the cytoplasmic TM1–TM2 linker, and 7 located in the cytoplasmic TM3–TM4 linker.
Phylogenetic Analysis
To understand the relationships among the Bdorα6 protein with other insect nAChRs, a phylogenetic tree was constructed using the neighbor-joining method (Fig. 2). Phylogenetic analysis indicated that the Bdorα6 was most closely related to Dα6 and Agamα6 with these 3 subunits clustering together, and also showed the relationships with different subunits from other species. Sequence BLASTP revealed that the deduced amino acid sequence of Bdorα6 was highly conserved with alpha6 homolog in other insect species. Compared with proteins in alpha6 subunit from the NCBI database, Bdorα6 shared 94.53% identity with the Dα6 of D. melanogaster (AAM13393), 87.78% identity with the Agamα6 of A. gambiae (AAU12509), 81.08% identity with the Bmα6 of B. mori (ABL67934), and 72.21% identity with Amelα6 of A. mellifera (AAY87894). Therefore, the cloned nAChR subunit gene should belong to α6-type.
Developmental and Tissue-Specific Expression Pattern of Bdorα6
Quantitative real-time PCR was employed to investigate the Bdorα6 gene expression levels during various developmental stages and different adult tagmatas. The result showed that the Bdorα6 gene was expressed in all stages, indicating that it has a role throughout the entire life cycle. The lowest mRNA level was found in pupa stage, and the relative expression levels of Bdorα6 was 23-, 39-, 6-, 2- and 27-fold higher in the egg, first, second and third instar larvae, and adult than in the pupa, respectively. Subsequently, the relative expression level of Bdorα6 in egg, second, third instar larvae, and pupa were significantly lower from that in the first instar larva (P < 0.05) (Fig. 3). There was a descending expression level of Bdorα6 during the developmental period from the first instar larva to the pupa.
The Bdorα6 mRNA was found in different tagmata of adult flies. The relative expression level of Bdorα6 was significantly different (P < 0.05) between different tagmata, and it was 109- and 15-fold higher in the head and thorax relative to the abdomen, respectively (Fig. 4).
Fig. 1.
Nucleotide and deduced amino acid sequence of Bdorα6 cDNA from Bactrocera dorsalis (GenBank accession number: JF974072). The start codon is indicated with bold and the stop codon is indicated both with bold and an asterisk. The predicated signal peptide is double underlined. The Cys-loop domain is underlined with a dotted line. Potential N-linked glycosylation sites are boxed. The position of putative 4 trans-membrane domains (TM1– 4) is shaded. The cysteine doublet (characteristics of nAChR α subunits) is marked with as ‘####’. The potential phosphorylation sites are underlined.

DISCUSSION
In this study, we identified and characterized Bdorα6 cDNA from the oriental fruit fly, B. dorsalis. Alignment of the deduced amino acid sequence with the nAChR homologs revealed that Bdorα6 possesses typical nAChR subunit characteristics, such as a signal peptide, a long N-terminal extra-cellular domain, and 4 hydrophobic transmembrane domains (TM1–4). Other important amino acid residues conserved in nAChRs were also identified. The N-terminal domain includes a cysteine loop consisting of 2 cysteines separated by 13 residues, which is the best-conserved feature in all ligand-gated ion channels, and the ligand-binding site forming regions (loops A–F) (Grutter & Changeux 2001; Karlin 2002). Additionally, because of the 2 adjacent cysteines in loop C, the proteins are defined as α-type subunit (Corringer et al. 2000). A BLASTP search against the NCBI protein database found that Bdorα6 sequence was highly similar to many other insect nAChR α subunits. With high similarity to the nAChR α6 subunits from A. mellifera, A. gambiae, and D. melanogaster, we deduce the Bdorα6 belongs to α6-type of nAChR.
Fig. 2.
Evolutionary relationships of deduced amino acid sequence of Bdorα6 with nAChR subunits of Anopheles gambiae and Drosophila melanogaster constructed using the neighbor-joining method. Bootstrap values with 1000 trials are indicated on branches. The scale bar represents the number of substitutions per site. Sequences (GenBank accession numbers) used: Anopheles gambiae Agamα1 (AAU12503), Agamα2 (AAU12504), Agamα3 (AAU12505), Agamα4 (AAU12506), Agamα5 (AAU12508), Agamα6 (AAU12509), Agamα7 (AAU12511), Agamα8 (AAU12512), Agamα9 (AAU12513), Agamβ1 (AAU12514); Drosophila melanogaster Dα1 (CAA30172), Dα2 (CAA36517), Dα3 (CAA75688), Dα4 (CAB77445), Dα5 (AAM13390), Dα6 (AAM13393), Dα7 (CAD86936), Dβ1 (CAA27641), CAD86936β2 (CAA39211), Dβ3 (CAC48166).

During development process, the expression of nAChRs changes considerably (Gotti & Clementi 2004). In D. melanogaster, the expression of Dα6 was most abundant in embryos and then decreased in both larvae and adults (Grauso et al. 2002). A similar developmental pattern in expression was found in rats and humans (Gotti & Clementi 2004). In adult L. bostrychophila, higher amounts of Lbα1 and Lbα8 transcripts were found compared to nymphal stages (Tang et al. 2009). Furthermore, the mRNA expression level of Pxα8 was higher at adult stage than at either pupal or fourth instar larval stage in P. xylostella using semi-quantitative RT-PCR (Zhao et al. 2009). In the present study, Bdorα6 was found to be present at all life stages, and most abundant in the first instar larva followed by adult, egg, second and third larval stages and the lowest in pupa. Therefore, the relative high expression level of Bdorα6 in the first instar larva was unique and its significance will require further investigation.
Fig. 3.
The relative expression levels of the Bdorα6 in different developmental stages of Bactrocera dorsalis (bar graph represents Mean ∓ SE; different letters above each bar indicate significant differences, P < 0.05, LSD in ANOVA).

The nAChRs are ligand-gated ion-channels expressed in many regions of the brain in insect. Several nAChR subunits transcripts from various species were detected in different tagmata. In the present study, Bdorα6 was found to be present in all tagmatas of adult flies the highest abundance in head followed by thorax and the lowest in abdomen. These results agreed well with the expression levels of nAChR genes Mdα2, Mdα5, and Mdα6 found in M. domestica (Gao et al. 2007a, b; Gao et al. 2007c). However, the highest levels of Accβ1 and Accβ2 mRNA were found in the abdomen of Chinese honey bee (A. cerana cerana) (Yu et al. 2011). The relative expression level of Mdβ3 in the abdomen of M. domestica was also higher than in the thorax (Gao et al. 2007c). These results indicate that different nAChR subunits may exhibit distinct functions in different species. In Drosophila, Dα2 transcripts and protein were exclusively in the CNS detected by in situ hybridization and immunohistochemistry, and suggested that the Dα2 protein was a subunit of a synaptic nicotinic receptor (Jonas et al. 1994). In the honeybee, Apisα3 mRNA expression was restricted to the suboesophageal ganglia in larvae, while it is further expressed in the optic, dorsal and antennal lobes, and the calyces of mushroom bodies in adults (Thany et al. 2003). While the B. dorsalis Bdorα6 mRNA transcript was most abundant in the head, and this pattern is consistent with the idea that Bdorα6 is expressed in the central nervous system (CNS). The CNS of adult B. dorsalis is highly specialized and has a complex of brain and suboesophageal ganglia in head, a fusion of the thoracic and abdominal ganglia in thorax, and mainly distributes in the head. However, the localized distribution in the head at different developmental stages remains unknown. Therefore, further experiments need to be performed to have additional evidence to confirm this result.
Fig. 4.
The relative expression levels of the Bdorα6 in different tagmata of adult Bactrocera dorsalis (bar graph represents Mean ∓ SE; different letters above each bar indicate significant differences, P < 0.05, LSD in ANOVA).

In summary, we first cloned and characterized the cDNA of a nAChR subunit, Bdorα6, and analyzed its gene expression in different developmental stages and in different tagmata of adult B. dorsalis. Resistance to insecticides has become a major worldwide problem, and mutations of insect nAChR subunits may lead to high level of resistance (Liu et al. 2005; Perry et al. 2007). Our results may provide clues for understanding of physiological, pharmacological properties and mechanisms of insecticide resistance. In addition, determining the sequence of the nAChR cDNA can provide a first step to examine mutations that may give rise to insecticide resistance. Future work should focus on the understanding of the roles of nAChR in comprehending the insecticide-resistance mechanism of B. dorsalis.
ACKNOWLEDGMENTS
This work was supported in part by the National Basic Research Program of China (2009CB125903), Natural Science Foundation of Chongqing (CSTC, 2009BA1042, JJA80020), the Program for Changjiang Scholars and Innovative Research Team in University (IRT0976), the Specialized Research Fund for the Doctoral Program of Higher Education (20100182120022), and the Earmarked Fund for Modern Agro-industry (Citrus) Technology Research System of China.