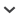The immense terrain of limestone karsts stretching from southern China to northern Vietnam (Sino-Vietnamese limestone karsts; SVLK) is a renowned biodiversity hotspot. It is noted for extremely high species diversity and endemicity of narrowly distributed calciphilous plants that are increasingly threatened by habitat destruction caused by rapid economic growth in recent decades (Chung et al., 2014). To design effective conservation strategies, a better understanding of the evolutionary mechanisms underlying the rich and distinct SVLK flora is essential. With ca. 70 species distributed exclusively in the SVLK, Begonia L. sect. Coelocentrum Irmsch. (Begoniaceae) is one of the most characteristic plant groups (Chung et al., 2014; Peng et al., 2014, 2015; Li et al., 2016), presenting an ideal model system for studying limestone plant speciation. Species of the section exhibit great morphological variation in leaf shape, texture, and variegation; they are usually confined to caves and cave-like microhabitats that occur abundantly in the karst areas of southern China (Guangdong, Guangxi, and Yunnan provinces) and northern Vietnam (Chung et al., 2014). Most species of sect. Coelocentrum are known from a single or a few localities, with the exception of B. cavaleriei H. Lév., B. leprosa Hance, and B. luzhaiensis T. C. Ku (Gu et al., 2007).
Based on phylogenetic analyses of Asian limestone Begonia species, Chung et al. (2014) proposed that the excess of moisture brought by the onset of East Asian monsoons since the late Miocene accelerated rates of karstification of Sino-Vietnamese limestone terrains, triggering widespread allopatric speciation in the SVLK. To test the microevolutionary processes underlying the speciation hypothesis of Chung et al. (2014), we investigate the population genetics and phylogeography of B. luzhaiensis. In this article, we report microsatellite markers developed for B. luzhaiensis. Expressed sequenced tag–simple sequence repeat (EST-SSR) markers are valuable in tests of cross-transferability, facilitating studies of population genetic diversity in many plant species (e.g., Dikshit et al., 2015; Zhou et al., 2016). Here, we used next-generation transcriptome sequencing to develop a set of microsatellite markers for B. luzhaiensis. Additionally, we tested the transferability of these markers for B. leprosa, another widespread species of sect. Coelocentrum.
METHODS AND RESULTS
To maximize potential loci, total RNAs were extracted from fresh leaves and male floral buds (from C.-I Peng 18732, denoted as Pool AC) and fruit (C.-I Peng 18735, Pool B) of B. luzhaiensis, respectively. RNA extraction was performed using the PureLink RNA Mini Kit (Invitrogen, Carlsbad, California, USA) according to the manufacturer's instructions, and quality and quantity were measured by an Agilent 2100 Bioanalyzer (Agilent Technologies, Santa Clara, California, USA). Illumina TruSeq library preparation and sequencing using the Illumina MiSeq system (2 × 250 bp paired-end; Illumina, San Diego, California, USA) were performed by Tri-I Biotech (New Taipei City, Taiwan). Reads were de-multiplexed, quality-trimmed, and assembled using SOAPdenovo2 (Luo et al., 2012). All sequence information has been deposited in the National Center for Biotechnology Information (NCBI) Sequence Read Archive (SRA; PRJNA378679). Our assembly generated 40,226 and 28,454 contigs for the Pool AC and Pool B assemblies. The average contig length was 874 and 850 bp for Pool AC and Pool B, respectively. In combined B. luzhaiensis assemblies (combined Pool AC and B), 44,368 contigs with an average length of 864 bp were generated.
Table 1.
Characteristics of 16 microsatellite loci developed for Begonia luzhaiensis.

The program Simple Sequence Repeat Identification Tool (SSRIT; Temnykh et al., 2001; http://archive.gramene.org/db/markers/ssrtool) was used to identify sequences containing at least five di-, tri-, tetra-, penta-, and hexanucleotide microsatellite repeats. In combined B. luzhaiensis assemblies, 5602 microsatellite-containing sequences were identified, of which 60 potential loci were selected for primer design using Primer-BLAST (Ye et al., 2012), with the optimum conditions set at a length of 20 bp (18–22 bp), a melting temperature of 60°C (57–63°C), and a product size range of 120–250 bp.
To characterize the degree of polymorphism of each locus, 57 individuals from three populations were genotyped using the 60 newly designed primer pairs (Appendix 1). Total genomic DNA was extracted from silica gel–dried leaves based on protocols outlined in Chung et al. (2014). The PCR reaction was conducted with a final volume of 20 µL containing approximately 30 ng of genomic DNA, 1 µL of 10 µM of each primer, and 10 µL of 2× Master Mix Red (Ampliqon, Odense, Demark). The following PCR conditions were used: an initial denaturation of 94°C for 5 min; 32 cycles of 95°C for 40 s, 53°C for 35 s, and 72°C for 1 min; followed by an extension of 5 min at 72°C. The amplified products were analyzed on an ABI 3500 Genetic Analyzer (Applied Biosystems, Waltham, Massachusetts, USA) with GeneScan 600 LIZ Size Standard (Applied Biosystems). Genotypes were determined using GeneMarker version 3.7 (Holland and Parson, 2011).
Of the 60 primer pairs, 16 loci were polymorphic among the three tested populations (Table 1). The number of alleles per locus, expected heterozygosity, and observed heterozygosity were calculated with GenAlEx 6.503 (Peakall and Smouse, 2012). GENEPOP 4.2 (Raymond and Rousset, 1995) was used to perform exact tests of Hardy–Weinberg equilibrium and linkage disequilibrium. The total number of alleles ranged from one to nine with a mean of 3.379 (Table 2). The observed and expected heterozygosity ranged from 0.000 to 1.000 and from 0.000 to 0.804 with averages of 0.370 and 0.404, respectively. Significant deviations of Hardy–Weinberg equilibrium in terms of heterozygosity deficiency were detected at three loci (BLZ01, BLZ06, BLZ16) in the Luzhai population (LZ, Table 2). Significant linkage disequilibrium was not detected between any pair of loci (P < 0.001). The putative functions of SSR-associated sequences were determined by BLASTX against the nonredundant GenBank database.
Table 2.
Genetic characterization of 16 newly developed polymorphic microsatellites of Begonia luzhaiensis.a

Table 3.
Cross-amplification results for the 16 microsatellites developed for Begonia luzhaiensis in seven populations of B. leprosa (n = 1).a

The interspecific transferability of the 16 markers was evaluated in B. leprosa, another widespread species of sect. Coelocentrum (Table 3). Five markers were successfully cross-amplified in B. leprosa.
CONCLUSIONS
The 16 microsatellite markers described here are the first developed for B. luzhaiensis and also the first study in Begonia sect. Coelocentrum. These microsatellites can be applied to the investigation of genetic diversity, population genetic structure, mating system, and gene flow, thus facilitating our understanding of evolutionary mechanisms and species diversification of the limestone flora. Data from such studies will contribute to the conservation and management of B. luzhaiensis that is increasingly threatened by habitat destruction.
ACKNOWLEDGMENTS
This work was supported by grants from the Biodiversity Research Center, Academia Sinica, Taiwan, and the Ministry of Science and Technology, Taiwan (NSC 101-2621-B-001-003) to C.I.P. and K.F.C.