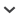Sphenophorus incurrens Gyllenhal (Coleoptera: Curculionidae) is an insect that feeds mainly on plants of the family Poaceae in the Neotropics. In Mexico, this weevil is an emergent pest of sugarcane. In this study, first the behavioral responses of both sexes to conspecifics were evaluated in a Y-tube olfactometer. Second, the volatiles of S. incurrens were sampled by the dynamic headspace technique and analyzed by gas chromatography coupled with electroantennographic detection (GC-EAD). Third, the antennal-active compound was identified by gas chromatography coupled with mass spectrometry. Finally, the biological activity of the identified compound was evaluated in laboratory and field tests. Our results showed that males emitted a pheromone that attracts both sexes. The GC-EAD analysis of the male volatiles showed that one peak elicited antennal responses from male and female weevils. The compound was identified as 2-methyl-4-octanol. Laboratory and field tests showed that the identified compound is attractive to both sexes of this weevil species, confirming its pheromonal activity.
Sphenophorus incurrens Gyllenhal (Coleoptera: Curculionidae) is an insect that feeds mainly on plants of the family Poaceae, such as sugarcane (Saccharum spp.), rice (Oriza sativa L.) and kikuyu grass (Pennisetum clandestinum Hochst. ex Chiov.) (van Zwaluwenburg 1926; Flores 1994; Ordaz-Gonzáles et al. 2014). This weevil is of Neotropical origin and has been observed from Mexico to Panama (Vaurie 1951, 1954; Anderson 2002).
In the past, this weevil was considered a sugarcane pest of minor importance in Mexico because its population levels were low. The beetles mainly attacked old plantations, and damage was restricted to small areas (van Zwaluwenburg 1926; Flores 1994). Currently, however, S. incurrens has acquired relevance because its populations have increased to a level that is causing economic damage in several sugarcane-producing states of Mexico (Pérez-De La O et al. 2014; Ruiz-Montiel et al. 2015). The main damage by this insect is caused by its larvae, which feed on the plant rhizome producing galleries that allow the entry of opportunistic pathogens that may cause plant death. So far, there is no effective control method for this insect pest; therefore, various control strategies would be worth exploring. One of these possibilities is the use of semiochemicals as a component of integrated pest management. A number of studies have reported the presence of pheromones and kairomones in several species of the subfamily Rhynchophorinae (Bartelt 1999; Piñero & Ruiz-Montiel 2012), to which S. incurrens belongs. These compounds may be used for monitoring pest populations or as a control method (e.g., mass trapping) (Wyatt 2014). There are some successful examples of the use of semiochemicals for monitoring, mass trapping, and mating disruption of weevil species (Dickerson et al. 1987; Chinchilla et al. 1996; Downham et al. 2001; Oehlschlager et al. 2002; Piñero et al. 2011).
In this study, we performed behavioral bioassays to determine whether this weevil species produces an aggregation pheromone (i.e., a semiochemical produced by a sex that attracts both sexes about equally). Second, we sampled the volatiles of S. incurrens by dynamic headspace technique and analyzed them by coupled gas chromatography— electroantennographic detection (GC-EAD). Third, we identified the electrophysiologically active compound by coupled gas chromatography— mass spectrometry (GC-MS). Finally, we evaluated the biological activity of the identified compounds in an olfactometer and field tests.
Materials and Methods
BIOLOGICAL MATERIAL
Adult weevils of unknown age and mating status were collected in damaged sugarcane plantations in the locality of Potrero, municipality of Atoyac, State of Veracruz, Mexico. Insects were taken to the laboratory, where they were sexed and placed in plastic containers (30 × 20 × 20 cm). Pieces of fresh sugarcane (10 mo old), variety CP-72 2086, harvested from commercial plantations were used for feeding weevils and for bioassays. The insects were maintained at 25 ± 2 °C and 70 ± 10% RH.
BEHAVIORAL RESPONSES OF S. INCURRENS TO VOLATILES FROM CONSPECIFICS AND SUGARCANE
The responses of S. incurrens females and males to volatiles of conspecifics and sugarcane were evaluated in a glass Y-tube olfactometer following the method described by Ruiz-Montiel et al. (2003). The olfactometer (stem, 12 cm; arms, 12 cm at 45°; internal diameter, 2.5 cm) was placed in an air-conditioned room (22 ± 2 °C, 70 ± 10% RH, and illumination 3 lux). Airflow was 0.5 L/min through each arm; the air was purified by activated charcoal and humidified before passing over the target and entering the Y-tube. Each arm was attached to a flow meter and an odor source container (a glass container 4.5 cm in diameter and 15 cm tall). The upwind ends of the Y-tube arms were each connected to the odor source containers, one of which contained one of the treatments, and the other contained the other treatment. A weevil, a male or female, was placed into the olfactometer base tube and given 5 min to walk toward the end of one of the arms. Choice for an odor source was defined as a weevil crossing a line 7 cm upwind of the bifurcation of the Y-tube and remaining there for at least 20 s. If a weevil did not make a choice within 5 min, this was recorded as no response. After each trial, the olfactometer was washed and dried at 120 °C to avoid contamination. The position of odor chambers to each arm was reversed after each replication to eliminate potential bias due to odor source location. For each odor source pair, 40 weevils of each sex were tested. The following treatments were compared: 1) males + sugarcane vs. clean air, 2) females + sugarcane vs. clean air, 3) males + sugarcane vs. sugarcane, 4) females + sugarcane vs. sugarcane, and 5) males + sugarcane vs. females + sugarcane. In all treatments, 15 males or 15 females and/or 100 g sugarcane were used as odor sources. Rectangular pieces of unpeeled sugarcane were used.
HEADSPACE SAMPLING OF WEEVIL VOLATILES
Volatiles emitted by both sexes of S. incurrens were sampled using the dynamic headspace technique. Typically, 100 females + 100 g sugarcane, 100 males + 100 g sugarcane, or 100 g sugarcane were placed separately into cylindrical glass aeration chambers (12.5 cm long × 4.8 cm inner diameter). The pieces of unpeeled sugarcane were cut longitudinally. A charcoal-filtered air stream (1 L/min) was maintained through the glass aeration chambers for 120 h. Volatiles were collected into glass traps (40 mm long × 4 mm inner diameter) containing 50 mg Porapak Q adsorbent (Alltech Associates, Deerfield, Illinois). At the conclusion of each air entrapment, the volatiles were eluted from the adsorbent with 400 μL methylene chloride (Baker, HPLC grade), and extracts were concentrated with N2 before analysis.
GAS CHROMATOGRAPHY—ELECTROANTENNOGRAPHIC DETECTION ANALYSIS (GC-EAD)
The GC-EAD analysis was performed to locate antennal-active components from Porapak Q extracts, using a Shimadzu GC-2010 plus gas chromatograph with a SPB-1 non-polar fused silica capillary column (30 m × 0.25 mm) (Supelco, Toluca, Mexico) programmed from 50 °C, maintained for 2 min, to 280 °C, at 15 °C/min. The EAD recording was made with an excised weevil antenna, and one antenna per weevil was used in each run. Both ends of the antenna were fixed between 2 EAG probes with electrically conductive gel (Spectra 360 electrode gel, Parker Laboratories, Fairfield, New Jersey). Antennal signals were recorded using GCEAD software version 2.6 (Syntech, Kirchzarten, Germany). The extracts of females and males were tested on 5 antennae per sex.
CHEMICAL ANALYSIS
The Porapak Q extracts were analyzed with a Varian CP-3800 GC linked to a Varian Saturn 2200 mass spectrometer (Varian, Palo Alto, California). The capillary column and the temperature program were the same as those described for the GC-EAD analysis. The carrier gas was helium at a constant flow rate (1.0 mL/min). The injector port temperature was held at 200 °C. The antennal-active component was tentatively identified based on comparison with spectra from the NIST/EPA/NIH Mass Spectral Library (version 2.0, 2002). The identity of the compound was confirmed by comparing retention times and mass spectra of synthetic standards. The synthetic 2-methyl-4-octanol was prepared according to procedures reported elsewhere (Perez et al. 1997). This compound was identified as a component of the aggregation pheromone of Metamasius hemipterus sericeus (Oliver) (Perez et al. 1997). Purity of the compound was 95% based on capillary GC analysis using a flame ionization detector.
BEHAVIORAL RESPONSES OF S. INCURRENS TO THE ANTENNAL-ACTIVE COMPONENT
The biological activity of the identified compound was evaluated in the Y-tube olfactometer described above. Serial dilutions of the test compound were prepared in methylene chloride to make 1, 10, and 100 ng/μL solutions. A standard aliquot (1 μL) of the selected concentration was pipetted onto a piece of filter paper (Whatman No. 1, Whatman, Maidstone, England), exposed 20 s to air to allow solvent evaporation, and introduced into one of the odor chambers of the olfactometer. A piece of filter paper loaded with 1 μL of solvent (control) was introduced into the opposite odor chamber. Also, each odor chamber of the olfactometer contained 50 g unpeeled sugarcane. After both test and control had been introduced into the odor chambers, a weevil, male or female, was placed into the olfactometer base tube and given 5 min to walk toward the end of one of the arms as described above. The position of odor chambers to each arm was reversed after each replication to eliminate directional bias. All concentrations were replicated 40 times for each sex. The bioassays were performed under controlled conditions: 22 ± 2 °C, 70 ± 10% RH, and illumination 3 lux.
FIELD TRIAL
Field evaluation of the identified compound was performed in an area of 7,200 m2 planted with sugarcane variety ITV 92-1424 in Potrero Nuevo, municipality of Atoyac, State of Veracruz, Mexico. The experimental site was chosen because previous technical reports and samplings showed that S. incurrens has been causing damage to the sugarcane plantations located in this area.
The traps used for the experiment consisted of white plastic containers with a capacity of 5 L (22 cm high × 20 cm diameter). Four 4 cm holes were made in the container wall to allow the weevil to enter the trap. The holes were made at a height of 5 cm from the container lid and 11.5 cm apart. Once the bait was introduced into the traps, these were buried, leaving the holes free to allow entrance of weevils. Traps were filled with 1 L of soapy water (1%) to capture the weevils attracted to the bait.
The following treatments were evaluated: 1) 250 g sugarcane alone, 2) 250 g sugarcane + 18 S. incurrens males, 3) 250 g sugarcane + a pheromone lure, 4) a pheromone lure alone, and 5) an empty trap as a control. The sugarcane was cut into small 5 cm long pieces. The pheromone lure consisted of a membrane device (Squid, Biological and Pheromones, S. A. de C. V. Texcoco, Mexico) loaded with 350 mg 2-methyl-4-octanol. According to the seller, this device releases about 1 mg per day. The membrane device was hung from a wire tied to two 0.5 cm holes at the center of the container lid. The sugarcane and males were placed in a plastic container (13 cm high × 11 cm diameter) with several holes in the lid to allow odor to disperse. The container was then placed inside the trap. The treatments were arranged in a complete random design with 4 replicates. The distance between traps was about 30 m. The experiment lasted 19 d, from 29 Apr to 17 May 2015. Traps were checked every other day, resulting in 9 observational dates, and the captured weevils were removed at each observational date. Sugarcane and weevils were changed 9 d after starting the experiment. The captured weevils were kept in plastic containers and transported to the laboratory to be sexed.
STATISTICAL ANALYSES
The behavioral responses of S. incurrens to conspecific volatiles and the identified compound were analyzed by a G-test with Williams' correction for sample size (Sokal & Rohlf 1995). Trap captures were analyzed by a 1-way analysis of variance (ANOVA), and means were separated by Tukey's test (a = 0.05). Before analysis, normality and homogeneity of variance were checked. A chi-squared test was used to analyze if there was a significant difference in the number of females and males captured by traps. Field data were analyzed with the statistical software SAS, version 9.0 (SAS Institute 2001).
Table 1.
Behavioral responses of both sexes of Sphenophorus incurrens to volatiles from conspecifics and sugarcane in a Y-tube olfactometer.

Results
BEHAVIORAL RESPONSES OF S. INCURRENS TO CONSPECIFIC AND SUGARCANE VOLATILES
Both sexes of S. incurrens significantly preferred the volatiles emitted by conspecific weevils + sugarcane when compared with clean air. In the same way, both sexes were more attracted to volatiles released by conspecific males + sugarcane than to sugarcane alone. However, neither sex showed any preference for volatiles from conspecific females + sugarcane or for sugarcane alone. Males were not more attracted to volatiles from males + sugarcane than to volatiles from females + sugarcane. In contrast, females preferred the volatiles from conspecific males + sugarcane over volatiles from conspecific females + sugarcane (Table 1).
GAS CHROMATOGRAPHY—ELECTROANTENNOGRAPHIC DETECTION AND GAS CHROMATOGRAPHY—MASS SPECTROMETRY ANALYSES
The GC-EAD analysis of Porapak Q extracts from S. incurrens males and females showed 1 male-specific compound that elicited antennal response in both sexes (Fig. 1A). The antennal-active component was identified by GC-MS as 2-methyl-4-octanol [GC-EI-MS m-z 126 (M+-18, 1%), 111 (1), 87 (38), 69 (100), 57 (14), 43 (53)] (mass spectrum match with 2-methyl-4-octanol). When 10 ng of synthetic 2-methyl-4-octanol was subjected to GC-EAD analysis, repeatable EAD responses were elicited by this compound (Fig. 1 B).
BEHAVIORAL RESPONSES OF S. INCURRENS TO THE ANTENNAL-ACTIVE COMPONENT
Neither sex showed preference for the synthetic 2-methyl-4-octanol or solvent at the concentration of 1 ng/μL. At the concentration of 10 ng/μL, females, but not males, were attracted to the synthetic compound, relative to the solvent control. Both sexes were attracted to 2-methyl-4-octanol compared with the solvent at the concentration of 100 ng/μL. When the attractiveness of 2-methyl-4-octanol + sugarcane was compared against sugarcane, males, but not females, preferred the synthetic compound over the control at the concentration of 10 ng/μL. At the highest concentration evaluated, females, but not males, were attracted to 2-methyl-4-octanol + sugarcane compared with sugarcane alone (Table 2).
FIELD TRIAL
The number of weevils captured (n = 106) was affected by treatment (F = 18.15, df = 32, P < 0.001). Traps baited with pheromone lure + sugarcane caught more weevils than any other trap (Fig. 2). There was no difference in the numbers of weevils captured among traps baited with sugarcane alone, sugarcane + males, and pheromone lure alone. The female-to-male sex ratio of weevils captured ranged between 1 in traps baited with sugarcane alone (χ2 = 0.04, df = 1, P > 0.05) and sugarcane + pheromone lure (χ2 = 0.07, df = 1, P > 0.05) to 0.54 in traps baited with sugarcane + males (χ2 = 1.81, df = 1, P > 0.05) and 0.60 in pheromone lure alone (χ2 = 0.82, df = 1, P > 0.05).
Discussion
In this study, we found evidence that S. incurrens males produce a pheromone that attracts conspecific females and males in a Y-tube olfactometer. We were able to identify the compound released by S. incurrens males, and the function of this compound as an aggregation pheromone was confirmed in the laboratory and field tests. The presence of an aggregation pheromone released by males has been reported in at least 17 species of Rhynchophorinae (Ambrogi et al. 2009). For instance, Rhynchophorus palmarum L. males were highly attracted to conspecific females and males in a 2-choice pitfall olfactometer, whereas females were not (Rochat et al. 1991). Also, field experiments showed that traps baited with males + palm stem caught more conspecific weevils than those baited with palm stem alone (Rochat et al. 1991).
Table 2.
Behavioral responses of both sexes of Sphenophorus incurrens to different concentrations of synthetic 2-methyl-4-octanol in a Y-tube olfactometer.

The GC-EAD analyses of male extracts showed that only 1 compound elicited response from female and male antennae. The finding that S. incurrens males produced only 1 pheromone compound agrees with findings in other Rynchophorinae, including Dynamis borassi (F.), Sphenophorus levis Vaurie, Sitophilus zeamais Motschulsky, Sitophilus oryzae (L.), and 4 species of Rhynchophorus (Ambrogi et al. 2009). In contrast, in another 9 species of this subfamily, the pheromone blend is composed of at least 2 compounds (Bartelt 1999; Ambrogi et al. 2009). For example, 3 compounds constitute the pheromone of the Australian population of the New Guinea sugarcane weevil, Rhabdoscelus obscurus Boisduval. Remarkably, the Hawaiian population of this species produced only 1 of the 3 compounds as its pheromone (Giblin-Davis et al. 2000). Scyphophorus acupunctatus Gyllenhal males release 4 attractive compounds (Ruiz-Montiel et al. 2008). However, the major pheromone component is sufficient to get captures comparable with those obtained with the quaternary blend (Rodríguez-Rebollar et al. 2012).
Our results showed that 2-methyl-4-octanol was the compound that elicited antennal response from S. incurrens females and males. This compound has been identified as part of the pheromone blend of Metamasius hemipterus (L.) (Ramirez-Lucas et al. 1996; Perez et al. 1997), the Australian population of R. obscurus (Giblin-Davis et al. 2000), and S. acupunctatus (Ruiz-Montiel et al. 2008). In contrast, this alcohol alone functions as an aggregation pheromone for the Hawaiian population of R. obscurus (Giblin-Davis et al 2000) and for S. levis (Zarbin et al. 2003). Interestingly, the 2 species of Sphenophorus have been found to use 2-methyl-4-octanol as their pheromone (Zarbin et al. 2003). As this alcohol is chiral, it is possible that the species use different enantiomers. However, in the case of S. incurrens, it would be necessary to determine the absolute configuration of the released compound. Zarbin et al. (2003) characterized the compound naturally released by S. levis as (S)-(+)-2-methyl-4-octanol. Although both weevil species use the same host plant species, they are geographically separated; therefore, there is no risk of cross-attraction between them. It would be interesting to identify the pheromone of other species of Sphenophorus to determine the diversity of pheromone compounds used for this group of weevils and how they differ from other related genera. Generally, methyl branched ketones and alcohols are used as pheromone components by weevils of the subfamily Rhynchophorinae, with the exception of Cosmopolites sordidus (Germar), which uses a bicyclic ketal as pheromone (Beauhaire et al. 1995). In this way, it is possible that the alcohol/ketone pheromone motif is highly conserved within the subfamily, but more species need to be studied to confirm this idea.
Fig. 2.
Mean (+ SE; N = 9) number of Sphenophorus incurrens weevils caught in traps baits with the different treatments. SC, Sugarcane; SC+P, sugarcane + pheromone; SC+M, sugarcane + males; P, pheromone. Columns with the same letter are not significantly different (a = 0.05; Tukey's test).

The evaluation of the synthetic pheromone in the laboratory showed sexually dimorphic responses of S. incurrens. We observed that females were attracted to the concentration of 10 ng/ìL whereas males responded to the concentration of 100 ng/ìL when the pheromone was evaluated alone. This observation suggests that females are more sensitive to the pheromone than males. Other studies showed that the attraction of female and male beetles may be affected by the quantity of the aggregation pheromone (Walgenbach & Burkholder 1986; Schlyter et al. 1987). For instance, S. zeamais females were attracted to synthetic pheromone from 1 ng to 1 μg whereas males had a slightly higher threshold, from 10 ng to 10 μg (Walgenbach & Burkholder 1986). However, in the case of S. incurrens, this pattern of response was inverted when the host volatiles were added to the pheromone; males responded to the concentration of 10 ng/ìL, and females were attracted to the concentration of 100 ng/ìL. A number of studies showed that host plants can influence the attraction of insects to their pheromones (Landolt & Phillips 1997; Reddy & Guerrero 2004; Dickens 2006; Liu et al. 2013). For example, more Dendroctonus valens LeConte females than males were attracted to 3-carene when this host volatile was evaluated alone. However, when the pheromone, frontalin, was added to 3-carene, a male-biased attraction was observed (Liu et al. 2013). The sexually dimorphic responses of S. incurrens suggest that the pheromone may have a dual function, functioning as sex attractant and aggregation pheromone, but future studies will clarify the role of this compound in the chemical ecology of this weevil species.
In the field trial, we found that the best treatment for capturing weevils was the combination of pheromone lure and sugarcane, even better than the treatment containing males and sugarcane. One possible explanation for the higher capture rates by the synthetic pheromone and sugarcane over the males and sugarcane maybe because the first treatment released a larger quantity of pheromone. Whereas male weevils emit the pheromone in nanograms, the baits released the synthetic pheromone in milligrams. The finding that the pheromone lure alone was less attractive than when sugarcane was added is not surprising because it is well documented with other species that food volatiles synergize the response of weevils to their aggregation pheromone (Giblin-Davis et al. 1996; Reddy et al. 2005; Tinzaara et al. 2007). For example, the addition of fermented banana tissue to pheromone traps increased captures of C. sordidus weevils by 50% compared with pheromone alone (Tinzaara et al. 2007). Traps baited with the combination of synthetic pheromone and food attractants caught more R. obscurus individuals than those baited with synthetic pheromone or food attractants (Muniappan et al. 2004; Reddy et al. 2005).
Two aspects may facilitate the use of the pheromone for monitoring or mass trapping S. incurrens. First, the results of the field trial suggest that this species is attracted to the racemic blend of the pheromone, as are other species of Rhynchophorinae. It has been shown that non-naturally occurring enantiomers or stereoisomers in synthetic pheromones are behaviorally benign, thus allowing the use of racemic blends for monitoring or mass trapping weevils (Giblin-Davis et al. 1996; Reddy et al. 2005; Ruiz-Montiel et al. 2008). The second aspect is that the S. incurrens pheromone is composed of a single component instead of a blend of compounds, eliminating the necessity of additional experiments to determine the effects of various component ratios. However, several aspects related to the lure (e.g., release rate and longevity) and trap design need to be optimized before the pheromone identified here may be used in any operational program to monitor or control S. incurrens.
In this study, we found that S. incurrens males release an aggregation pheromone. Additionally, we identified the pheromonal component that was active in laboratory and field tests. Future studies will concentrate on optimizing the lure and identifying the food volatiles that synergize the response of weevils to this pheromone.
Acknowledgments
We thank Antonio Santiesteban (ECOSUR) for laboratory assistance, the staff, especially Arturo Martinez, at Fideicomiso Ingenio El Potrero S.A. de C.V. for facilities and assistance in the field experiment, and Squid, Biological and Pheromones S. A. de C. V. for providing pheromone lures and traps used in this study. This work was supported by CONACYT through a graduate scholarship to C. P. I. R.