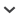Herein we describe the nests (including construction, closure, orientation, and depth of cells) of the bee Osmia (Ozbekosmia) avosetta Warncke found nesting near Antalya, Turkey, and Sepidan, Iran. Cells are unusual in that they are lined by two layers of colorful flower petals that sandwich a thin middle layer of mud. Analyses of pollen taken from scopal hairs of specimens from the Turkish site were identified as solely from Onobrychis viciifolia Scop. (Fabaceae) whereas those from the Iranian site were from a related plant, Hedysarum elymaiticum Boiss. and Hausskn. These facts coupled with analyses of scopal pollen from 11 other sites in Turkey, Jordan, and Syria strongly suggest that this bee is oligolectic with respect to the plant tribe Hedysareae.
The egg and last larval instar of Osmia avosetta are described. The presence of an egg taken from a cell and provisionally identified as belonging to Sapyga pulcherrima Morawitz suggests that this cleptoparasite may have this bee as one of its hosts.
In addition, we report new information on and review published accounts concerning the use of whole petals or large petal pieces in the construction of cell walls of osmiine bees. Only Osmia (Ozbekosmia) avosetta and species of Osmia (Tergosmia) have three-layered cell walls with the middle layer made of mud. Recorded also are the similarities and differences exhibited in pollen and petal preferences and nest characteristics of species in these two related subgenera.
INTRODUCTION
We present for the first time information about the nesting biology, floral preferences, and immature stages of Osmia (Ozbekosmia) avosetta Warncke, 1988. Although an uncommonly encountered species, two nesting aggregations of it were discovered almost simultaneously, one in Turkey and the other in Iran, and provided information for the current study. Additionally, we review the use of petals as a building material in osmiine bees and compare the biology of O. avosetta with that of species belonging to Osmia (Tergosmia). These two subjects are dealt with in the Discussion section.
The Turkish research party consisted of the first three authors (Rozen, Özbek, and Ascher), and the Iranian party of the next three (Sedivy, Praz, and Monfared). Müller was responsible for the study of floral preferences, the review of petal usage among osmiine bees, and the section Comparative Biology of Ozbekosmia and Tergosmia, as Rozen was for descriptions of immature stages.
DESCRIPTION OF NESTING SITES
In Antalya Province, Turkey, the Turkish party discovered two nests of Osmia avosetta at Seklik Mevkii (N 35°52′52″ E 30°22′30″), a small village about 6 km east of Saklıkent, the first nest on May 31, 2009, and the second on June 1, 2009. The elevation was approximately 1500 m on the east-facing slope of the mountain. The two nests were about 100 m apart, each on a small, partly to mostly barren mound of earth fully exposed to the sun. In each case, the nesting surface sloped 20°–40° from horizontal and the soil was moderately fine, loosely compacted, and friable. On June 3, 2009, we returned and found an aggregation of nesting females about 100 m from the closest nest site found earlier. It was at a slightly higher elevation, but still on the eastern slope of the mountain and fully exposed to the sun. The ground was moderately covered with stands of the food plant Onobrychis viciifolia Scop. (Fabaceae) between which were more thinly vegetated areas where we found nest entrances (fig. 1). The slope of the surface varied here from 0°–20°. The surface soil was crusty with pebbles and numerous fractures; beneath, the soil was dry and generally easily excavated, though with some rocks.
Fig. 1
Nesting site of Osmia avosetta in Seklik Mevkii, Turkey, to the left of observers in fore- and midground; note pinkish-red flowers of pollen plant, Onobrychis viciifolia. Figures 2, 3. Closed brood cells of O. avosetta, side views, showing shape and variation in coloration of outer envelopes. Figure 4. Open cell of same, side view. Figure 5. Closed cell of same containing intermediate stage larva. (Photos J.G. Rozen.)

The Iranian party discovered five nests of Osmia avosetta, which is a new species record for Iran, on May 31, 2009, along Yasouj-Shiraz new road, 6 km after Tange Tizab, at 10 km northeast of Sepidan, Fars Province, Iran (N 30°20′6″ E 51°50′22″, elev. 2270 m) (fig. 6). The nesting site was a large, very steep scree slope exposed to the north, with only scarce vegetation. The food source, Hedysarum elymaiticum Boiss. and Hausskn. (Fabaceae) (fig. 7), grew abundantly at the site and was visited by numerous females of Osmia avosetta, though no males were found. Although the ground at this site was mainly composed of stones and gravel, all nests were located in soil that was so loose and friable that we were able to dig out the nests by hand. Where soil was more compact on less sloping surfaces, no nests were found. The five nests were scattered, and each contained only one cell.
Figs. 6–9
Nesting of Osmia avosetta at 10 km northeast of Sepidan, Yasouj region, Iran. 6. The site. 7. Pollen plant, Hedysarum elymaiticum. 8, 9. Two brood cells. (Photos C. Sedivy).

The first nest was found because we saw a female enter a hole in the ground. This nest was still open, and the bee was inside the brood cell. We found four more nests (each containing a completely closed brood cell) by simply digging around other holes in the ground, possibly an indication that the females do not fill the burrows with pebbles after finishing the brood cell.
At the Turkish site, the entire nesting area was extensive, about 30 m long and 25 m wide. Some nest entrances were circular holes, 5–7 mm in diameter, and others were irregular in shape, often associated with the surface cracks (fig. 10). They were not uncommon (about 1/m2), but irregularly distributed. Both males and females were seen at the nesting area visiting flowers of Onobrychis or landing briefly on the ground in barren areas. Several tumbling pairs observed from a distance were probably mating, thus explaining the appearance of both sexes in the area.
Fig. 10
Female of Osmia avosetta with a petal in its mandibles approaching nest entrance (photo J.S. Ascher). Figures 11–15. Cells of Osmia avosetta. 11. Open cell, side view. 12. Close-up of neck of same, showing sequence of plating of petals. 13. Closed cell, side view. 14. Top of same showing inward folded petals of outer envelope. 15. Same cell, now with top of outer envelope removed revealing soil closure and soil lining, and some inner petal lining where soil lining removed (arrow). (Photos J.G. Rozen).

NEST ARCHITECTURE
Since it was our last day in the field at the Turkish site, we hurriedly excavated approximately 10 nests, which we recognized by entering or departing bees. Only two or three consisted of two cells. All others contained a single closed cell, an open cell being provisioned, a cell being constructed, or no cell. At first we interpreted the paucity of cells to indicate that the foraging season was only starting. However, in hindsight we realize that, if completed nests normally contain only single cells or two cells, we may well have overlooked numerous completed nests because all but one nest studied were initially identified by adults entering or leaving. Had we had one more day to identify and excavate nests lacking evidence of adult activity, conclusions might have been different. Indeed, we now tentatively conclude that they normally consist of one or two cells because most of the females collected at the time of our fieldwork bear lightly to moderately worn wings rather than unworn wings. Nests at both sites were shallow, with cells ranging in depth from 1.5 to at most 5.0 cm. In addition to the nest entrance, each had an open main burrow, 6–7 mm in diameter that in most cases descended vertically, but in one case slanted substantially to one side, presumably because the female had encountered a rock. There was no special burrow lining nor did the burrow wall seem consolidated in any way, such as by tamping.
CELL ORIENTATION AND STRUCTURE
Cells at both the Iranian and Turkish sites were mostly vertical or nearly so, although a number tilted as much as 10°–20° from vertical. Although only single cells were found in Iran, where two cells were found in a single nest in Turkey, they were normally positioned side by side and contiguous, with one cell several millimeters higher than the other. However, in one nest the lower cell met the side of the other at an angle.
Cell structure, also identical at the two sites, is complicated, as must be the stereotypic procedure used by the female bee to construct it. In describing cells of most bees, the inner dimensions (length and maximum width) of the cavity are generally presented with some idea of the shape of the lumen, but the cell of this species is delineated by the external shape of the lining and characterized by the color and materials that make up the lining. All materials used are collected and transported by the female from outside the nest. After the female digs the main tunnel and cavity for one or two cells, she brings in large pieces of petals to line what will become the outer envelope of the cell lining, probably first applying them to the lower surfaces of the cavity, and with successive importations advancing the lining upward on all sides, as evidenced by the shingling of petals on the cell neck (fig. 12). The outer envelope is extended into the neck of the cell, well beyond (about 3 mm) what will become the top of the closed cell. Later, as the last act of cell construction, the female will fold these petals mesad to the long axis of the cell to form the exterior cell closure of the completed cell (fig. 14). Apparently because petals of a wide range of available flowers are used, the colors on some cells include purple, blue, tan, yellow, and red (fig. 11–13), but on other cells there is a more limited array of colors (figs. 8, 9), probably indicating a narrower selection of appropriate plants in flower.
After the female completes the outer envelope, she then brings in fine moist soil and plasters it against the inner surface of this lining, forming a layer 0.5–0.8 mm thick. This layer covers the entire inner surface of the outer envelope to where the closure will be placed, so that the upper 3 mm of the petal lining remains uncovered. The source and nature of the moisture in the soil is unknown but may be water, a secretion, or possibly nectar. When the moist soil sets, it becomes dull, pale, and hard (cause of the setting unknown, but perhaps simple drying). As the next step, the female imports petals to completely line the cell cavity and thus forms the inner envelope, which extends upward beyond the soil lining. At the entrance the inner and outer envelopes adhere, forming a central passageway through which the female imports provisions and deposits her egg on the top surface of the provisions (fig. 21). Thus, the cell wall of the brood chamber before closure, except for the upper 3 mm, consists of three closely appressed layers: an outer envelope of petals and an inner envelope of petals that together sandwich a layer of soil.
Figs. 16–21
More closed cells of Osmia avosetta. 16. Upper part of cell, side view, with petals of outer envelope partly removed to reveal inner soil lining capped with closure of soil with concave upper surface. 17. Close-up of same showing concave soil closure, top view. 18. Same cell, now with most of soil closure removed revealing some of petals of inner closure (arrow), top view. 19. Same, except now all of soil of inner closure removed revealing folded petal of inner envelope, top view. 20. Another cell with outer envelope teased open, soil closure removed, and inner envelope closure also teased open, top view. 21. Same cell with opening widened to reveal egg floating on provisions. (Photos J.G. Rozen).

At the Turkish site, we assumed that the petals were harvested from the numerous adjacent flowering plants, including Onobrychis (fig. 1), as reflected in the multicolored outer envelopes of the cells. However, at the Iranian site the deep hue of the outer envelope (figs. 8, 9) did not appear at first to match any of the close-by flowering plants including the pale-flowered Hedysarum elymaiticum (fig. 7), the larval food source for Osmia avosetta at this site. Subsequently C.S. concluded on reexamining the cells that all petals seemed to originate from the banner petals of this plant and that their darker, more purple hue resulted from withering after the petals are cut. He noted that the petals were all shaped like the upper part of a heart and were arranged in the same manner: their tips pointed downward and the cut side pointed upward and they overlap like scales in both the inner and the outer petal linings.
After egg laying comes the task of closing the cell, a process that the female carries out in three steps. She first closes the inner envelop by folding mesad the apical petals of the inner envelope, thus closing the cell lumen and forming a truncated top for the inner envelope. This top measured about 4.5 mm in diameter (figs. 18, 19) in one case. She imports no additional petals from the outside. The closure petals are somewhat moist and can be teased apart and unfolded with forceps to expose the cell lumen below (fig. 20). The ease by which we were able to unfold the inner closure without tearing the petal tissue suggested the ease the female bee had in folding the soft and compliant petal tissue when constructing the closure.
As the next step she brings in a small quantity of dry soil to place into the slight concavity formed on the top of the inner envelope closure, followed either by importing moist soil or by adding some liquid to the dry soil to manufacture a smoothly concave outer (upper) surface to the soil closure (figs. 15–17). This closure, similar in texture and thickness to the soil lining of the cell wall, bonds at its periphery to the soil lining. How she manufactures the smooth surface to the concavity remains unknown.
As the final step to protect her offspring, the female folds mesad the petals at the open end of the outer envelope against the soil closure (fig. 14). On at least three closures, the females brought in and added several petals that were not part of the lining, presumably to augment the thickness to the top of the closure.
In Turkey a single closed cell was accidentally encountered when we were excavating another nest. It contained an intermediate-stage larva actively feeding on provisions. The colorful display of petals persisted on the outer envelope (fig. 5), but the soil lining to the cell had hardened considerably compared with this lining in fresher cells. Soon after being collected the larva molted to the last larval instar, described below. This accidental discovery added to our suspicion the nests contain only one or two cells, because this was the only larva that we found in the 10 or so nests we excavated at the Turkish site.
Cells still open from both sites were elongate (figs. 4, 11), because the visible lining included both the cell itself and the petal-lined neck. They measured 18.0–19.5 mm in length (N = 5). Closed cells were shorter since the linings of the neck regions are folded over and incorporated into the cell closure, as described above. Closed cells measured 15.0–17.5 mm (N = 9) in length. All cells (open and closed) were 7.0–9.0 mm in maximum outside diameter (N = 7). The widest part of closed cells was between one-quarter and one-third of the distance from the bottom of the cell. Cells were not symmetrical around their long axes; all had one side that was convex while the opposite side was slightly concave (figs. 2, 3).
Provisions were a sticky mixture of yellow-orange pollen, homogeneously combined with nectar. They occupied roughly the lower half of the inner envelope of petals and conformed to the shape of the bottom of the cell. On their surface, the female deposited her elongate, shiny egg (fig. 21), described below.
FLORAL PREFERENCES
At the nesting site near Saklıkent, Turkey, females of Osmia avosetta were seen visiting only the flowers of Onobrychis viciifolia Scop. (Fabaceae) for larval provisions, although many other flowers were available as potential pollen and nectar sources, e.g., Anchusa, Salvia, Centaurea, Astragalus, and Melilotus. The microscopic analysis of pollen contained in the abdominal scopa of nine females collected at the nesting site on June 3, 2009, revealed that all females had indeed harvested pollen exclusively from Onobrychis viciifolia.
At the nesting site near Sepidan, Iran, the females of Osmia avosetta were observed to exclusively visit the flowers of Hedysarum elymaiticum, although many other plants were flowering in the proximity of the nesting site, including several Fabaceae species of the genera Astragalus, Lotus, and Trifolium. Microscopic pollen analysis corroborated the narrow host-plant preference of Osmia avosetta at the Iranian site: pollen masses removed from the abdominal scopa of 15 females collected at the nesting site on May 31, 2009, were entirely composed of Hedysarum pollen as were the pollen provisions of five brood cells dug out the same day.
The exclusive utilization of pollen of Onobrychis and Hedysarum, which form a well-supported clade within the tribe Hedysareae (Wojciechowski et al., 2004; McMahon and Sanderson, 2006; Ahangarian et al., 2007), indicates that Osmia avosetta is most probably a specialist on this plant tribe. This conclusion is supported by microscopic analysis of scopal pollen contents of 13 additional females collected at 11 different localities in Turkey, Syria, and Jordan, which were all exclusively composed of tricolpate Fabaceae pollen typical for Onobrychis and several Hedysarum species as well as for two other genera of the Hedysareae (Choi and Ohashi, 1996).
PARASITISM
No cleptoparasites were seen entering any of the nests at either site, but we retrieved from a cell a single egg unlike that of Osmia avosetta at the Turkish site. It differed by being slightly shorter (2.5 mm long), much thinner (maximum diameter 0.54 mm), faintly curved, and broadly rounded at the anterior end but tapering posteriorly and very narrowly rounded apically (fig. 31) with the widest part in the anterior one-quarter. We were unsuccessful in viewing its micropyle with an SEM. White in color like the host egg, it was suggestive of the egg of Sapyga luteomaculata Pic (22Rozen and Kamel, 2009), which is faintly tan, shorter (length 1.96–2.1 mm), and slimmer (maximum diameter 0.44–0.46 mm). The egg of S. louisi Krombein is similar though shorter (1.5 mm long) and narrower (0.25 mm at middiameter) (Mathews, 1965: fig. 16). The described egg of S. pumila Cresson (Torchio, 1972: figs. 2, 4) is even shorter (1.3 mm long) and, proportional to its length considerably slimmer (diameter at anterior end: 0.18 mm). The slender, distinctive torpedolike shape of all previously described eggs strongly suggests that the parasitic egg found in the cell of O. avosetta is a Sapyga or some related Sapygidae. Because two individuals of Sapyga pulcherrima Morawitz, 1894, were collected (but not entering nests) at the nest site during our study, it likely is that species.
IMMATURE STAGES
Immatures of Osmia avosetta and the egg of Sapyga pulcherrima described were collected at Seklik Mevkii, 6 km east of Saklıkent, Antalya Province, Turkey, June 3, 2009 (J.G. Rozen).
Description of Egg
Diagnosis
The egg of Osmia avosetta appears unremarkable, although we were unable to examine it with an SEM and therefore cannot describe its micropyle.
Description
Length 2.93 mm (N = 2); approximate maximum width 0.93 mm (N = 3). Egg index 0.81 (see Remarks). Upper surface slightly curved in lateral view (fig. 30); ventral surface nearly straight; anterior end (identified by developing embryo) slightly more narrowly rounded than posterior end in lateral view (fig. 30); sides subparallel when viewed from above; micropyle not visible with stereomicroscope. Egg color white; chorion under stereoscope clear, shiny, glassy, thin throughout.
Remarks
The egg index of 0.81 was calculated by dividing the average lengths of two eggs by the distance between the outer rims of the tegulae of a female from which one of the eggs was collected (Iwata and Sakagami, 1966). This value falls well within the medium category of Iwata and Sakagami's (1966: table 2) classification of bee egg size relative to female body size. This is the second species of osmiine bee with an egg index reported in this category; Hoplitis (Hoplitis) monstrabilis Tkalců has an egg index of 0.77 (Rozen et al., 2009).
Description of Last Larval Instar
The single larva recovered was an intermediate instar that molted to the last instar soon afterward. Last instars of larvae of probably all Megachilinae (except for the Lithurgini) can be recognized because of extensive body setae, which are absent on earlier instars (Baker et al., 1985; 22Rozen and Kamel, 2009). The larva was preserved after it had started defecating and spinning silk. Although a considerable amount of uneaten provisions remained in the cell, most fecal material had been voided by the time of preservation. Head capsule pigmentation was evident although whether it was at its maximum development is unknown. After being cleared by boiling in an aqueous solution of sodium hydroxide, pigmentation was greatly reduced.
Diagnosis
As was pointed out earlier, larvae of the Megachilidae are “very homogeneous” (Michener, 1953: 1040). Since then mature larvae of other megachilid taxa have been described (see McGinley, 1989, and references therein; Baker et al., 1985; Rozen and Özbek, 2004; Rozen and Kamel, 2007, 222009; and Rozen et al., 2009). From these works, we see that the Osmiini, Anthidiini, Dioxyini, and Megachilini as mature larvae can be easily distinguished from those of the more basal Pararhophitini, Fideliini, and Lithurgini because the former have conspicuous body setae and the latter do not. However, we still do not know how to recognize the tribes of the more advanced megachilids on the basis of their mature larvae, much less to distinguish larval Osmia from other Osmiini. Because the osmiine Hoplitis monstrabilis has approximately five setae on the pleural swelling of abdominal segment 8 (Rozen, et al. 2009), in contrast to approximately 80 in O. avosetta has and perhaps also the same number in O. (O.) ribifloris Cockerell, we at first thought that setal abundance might prove valuable. However, the larva of O. (Pyrosmia) submicans Morawitz from Egypt has only 12 setae on this swelling, suggesting that setal abundance may prove unreliable for generic recognition after more species are surveyed. Details of mandibular and atrial morphology seem to vary among the Megachilinae and may prove helpful in recognizing tribes and/or genera despite the high degree of larval homogeneity.
The peculiar absence of a sclerotized cardo in the presence of a well-sclerotized stipital rod is a feature unknown in several other mature larval Osmia that we have examined.
Description
Head (figs. 33, 34, 37): Setae long and abundant; those of frons shorter and those of labrum very short; those of maxillary and labial apices straight, forward projecting. Following areas moderately to faintly pigmented: labrum including labral sclerite (exclusive of middle part), darkest part of which is along subapical row of sensilla; area apicad of darkest part also sclerotized and pigmented, but extreme apical labral margin unpigmented, nonsclerotized; mandibles especially at apices and points of articulation; internal head ridges at articulation with mandibles; dorsal surface of premental sclerite between attachment of articulating arms of stipites; antennal papilla and all palpi only faintly pigmented. Fine spiculation restricted to dorsal surface of maxilla and lateral lobes of hypopharynx. Area immediately above hypostomal ridge and just behind posterior mandibular articulation not produced as downward-directed tubercle as present in many Coelioxys (Rozen and Kamel, 2007: fig. 47). Coronal ridge nearly absent; postoccipital ridge well developed; hypostomal ridge well developed, giving rise to pronounced dorsal ramus that extends posteriorly from middle of ridge nearly to postoccipital ridge (fig. 33) where it abruptly stops; anterior tentorial pit approximately equally distant from anterior mandibular articulation and basal ring of antenna; epistomal ridge present only laterad of anterior tentorial pits; tentorium robust including dorsal arms. Parietal bands faintly evident. Diameter of basal ring of antenna somewhat less than twice distance from ring to center of anterior tentorial pit; antennal papilla (fig. 39) small, slender, gradually, evenly tapering apically, about three times as long as basal diameter, bearing two to three sensilla (fig. 39). Lower margin of clypeus strongly angled upward at midline (fig. 34), so that at midpoint margin nearly at level of anterior tentorial pits. Labral sclerite transverse, pigmented (except at midline), with lower margin extending beyond apical band of sensilla; labrum lacking darkly pigmented median spot extending from labral sclerite to apical labral margin as in fully pigmented Coelioxys larvae (Rozen and Kamel: 2007: figs. 44, 45); apical labral margin moderately broad, distinctly concave (figs. 34, 37).
Mandible (figs. 35, 36) moderately robust; apex bidentate with ventral tooth longer than dorsal tooth; both teeth acutely pointed; dorsal apical edge of dorsal tooth finely, regularly crenulated; ventral apical edge of upper tooth and both edges of ventral tooth unmodified; apical concavity pronounced, sharply defined basally; cuspal area not developed; outer surface without setae or tubercles. Cardo as a sclerite absent; stipes a slender sclerotized rod posteriorly ending abruptly at point where it would have articulated with cardo; articulating arm of stipes evident; maxillary palpus moderately small, about same size as antennal papilla and labial palpus. Labium clearly divided into prementum and postmentum; apex normally wide (figs. 34, 37); premental sclerite weakly sclerotized, most evident dorsally; postmentum nonsclerotized. Salivary lips projecting, transverse, width about equal to distance between bases of labial palpi; inner surface of at least upper lip, visible only after specimen subjected to critical-point drying process, with numerous parallel, raised ridges extending outward (fig. 40). Hypopharynx consisting of two widely separated lateral lobes that are spiculate.
Body (figs. 32, 41): Body setae short, rising from swollen bases, abundant dorsally and laterally; pleural area of abdominal segment 8 with approximately 80 setae (fig. 41); integument in areas without setae with patches of very fine spicules. Body form robust; intersegmental lines weakly incised on predefecating larva; intrasegmental lines not evident but possibly visible on postdefecating form; paired body tubercles absent; middorsal body tubercles very evident on midbody segments, decreasing in size posteriorly (fig. 32); pleural swellings moderately developed; abdominal segment 10 attached to approximate middle of segment 9; anus positioned toward top of segment 10. Spiracles well sclerotized, unpigmented, subequal in diameter; atrium globular with width considerably greater than depth, projecting above body wall, with rim; peritreme narrow, so that diameter of atrial opening as much as four times peritreme width; atrial inner surface with rows of wrinkles (figs. 42–44) concentric with primary tracheal opening; atrial wall also with fine, sharply pointed, concentrically directed spicules; primary tracheal opening with collar; subatrium variable in length, with from four to 10 chambers; externally, subatrium tapering in side view. Male with small crescentic (almost circular) median integumental scar on venter of posterior edge of abdominal segment 9; female sex characters unknown.
DISCUSSION
Biology of Osmia avosetta
In light of the hazards that confront a nest shallowly situated in loose soil on a slopping surface, we can speculate as to how a cell with this construction might be beneficial. With little or no rain during the summer months, rainfall leading to soil erosion is not a significant threat until after the immature reaches hibernation in its cocoon. However, if the cell should be flooded by an unexpected early storm, the double petal lining would seemingly deflect much water, and the air trapped by two layers of petals would help float a cell that was eroded from the ground.
Probably a more significant hazard is desiccation especially in very shallow nests; the double layer of petals, which are a source of moisture in themselves, should help retain moisture during larval development. Because megachilid larvae in general seem little bothered by water loss, we wonder whether maintaining the water content of the provisions might be the selective force for the evolution of this elaborately structured cell wall. The rigidity added by the soil lining, of course, protects the cell contents from being crushed or invaded by predators and parasites.
Although the patchwork of colors on the outer surface of a cell (figs. 2–5, 11) or even the strong colors (figs. 8, 9) is a striking phenomenon to the human eye, color of the cell surface is obviously not important to the female bee or her nest. We think the survival value of constructing this elaborate cell lining of petals and soil is the texture, water content, and water repellent– and humidity-retaining nature of petals. High water content provides moisture, the soft tissue of petals makes them easy for the females to harvest, the compliant nature of the tissue is easily folded, and the lack of plant hairs allows petals to adhere to one another.
We here have interpreted the closure of the brood cell of Osmia avosetta as an integral component of the lining of the cell. Still to be explored is the construction of the cell closures of other osmiine taxa that use whole petals or large petal pieces to form cell walls. In Hoplitis (Anthocopa) dalmatica (Morawitz), whose cells consist of an outer layer of roughly chewed leaves and an inner layer of large petal pieces, the females immediately close the cell after egg deposition by folding mesad the apical petals (Müller, Krebs, and Amiet, 1997), corresponding to the first step of cell closure in Osmia avosetta. With newly collected leaf material that is applied onto the petal layer, H. dalmatica then finally closes the brood cell.
Petals as Nest Building Material in Osmiine Bees
Among the osmiine bees, the use of petals for brood cell construction is not confined to Osmia avosetta. Roughly chewed petals of Dalea (Fabaceae) are used by Ashmeadiella (Isosmia) rubrella Michener to build entire brood cells (Yanega, 1994), and some osmiine bee species, which usually utilize chewed green leaves as nest-building material, occasionally also use chewed petals, e.g., Hoplitis (Dasyosmia) biscutellae (Cockerell) (Rust, 1980), Osmia (Allosmia) rufohirta Latreille (Grandi, 1961), or O. (Helicosmia) caerulescens (Linnaeus) (Westrich, 1990). However, the use of whole petals or large pieces of petals for brood cell construction is restricted among the Osmiini to O. (Ozbekosmia) avosetta, two species of Wainia (Caposmia), most Hoplitis species of the subgenus Anthocopa as well as all Osmia species of the subgenus Tergosmia whose nesting biology has been recorded so far (Müller et al., unpubl.). In the two Wainia species, which both nest in empty snail shells, the partitions between the brood cells are constructed from large petal pieces glued together, whereas the nest plug is built from petals followed by a thick layer of cemented sand (Gess and Gess, 2008). The brood cells of Hoplitis (Anthocopa), which are generally built in excavated or preexisting burrows in the ground or, more rarely, in fissures and holes in rocks and bark, under stones or in empty snail shells, are entirely constructed from foreign material (Müller et al., unpubl.). Petals are used by most of these species as the exclusive cell building material. However, the two closely related H. (Anthocopa) graeca (Tkalců) and H. (Anthocopa) villosa (Schenck) apply mud in varying quantities to cement the petals together (Ducke, 1900; Friese, 1923; Petit, 1970; Westrich, 1990; A. Müller, personal obs.). In contrast to O. avosetta, however, the cells of these two species are not three-layered; instead petals and mud are more thoroughly mixed. A three-layered cell structure consisting of a mud layer sandwiched between two layers of petals, which is very characteristic of O. avosetta, is also found in O. lunata Benoist (A. Müller, personal obs.), O. rhodoensis (Zanden) (C. Praz and C. Sedivy, personal obs.) and O. tergestensis Ducke (Ferton, 1897; Müller et al., 1997), which all belong to the subgenus Tergosmia. This unique cell structure shared by both Ozbekosmia and Tergosmia indicates that these two subgenera most probably form a monophyletic group within the genus Osmia. This hypothesis is supported by the close morphological similarity of O. avosetta to Tergosmia, e.g., in the form of the female clypeus and of the male sterna (Michener, 2007). Indeed, Warncke (1988) placed O. avosetta in the subgenus Tergosmia before Zanden (1994) moved it to the new subgenus Ozbekosmia due to several deviating characters not found in Tergosmia. A recent molecular phylogenetic analysis of the Osmiini, which did not include O. avosetta, placed the three petal-using osmiine bee taxa Wainia (Caposmia), Hoplitis (Anthocopa), and Osmia (Tergosmia) in three distantly related clades (Praz et al., 2008). Thus, assuming that Ozbekosmia + Tergosmia together are indeed monophyletic, the use of whole petals or large petal pieces for brood cell construction has independently evolved at least three times within the osmiine bees.
The advantage of using large petals for brood cell construction instead of chewed green leaves (“leaf pulp”), which is a considerably more widespread nest-building material among the osmiine bees (Müller et al., unpubl.), remains unclear. The potential benefits of leaf pulp, e.g., chemical protection of the provisions by antimicrobial substances contained in the leaves, humidity control within the cell, or prevention of the absorption of nectar by the substrate, are expected to be properties of petals as well. One advantage of the use of large petal pieces over that of leaf pulp might be more efficient collection and processing, as the latter can be collected only in small quantities on a single flight. All petal-using osmiine bee species, for which petal collection has been recorded to date (Westrich, 1990; A. Müller, personal obs. for several Hoplitis (Anthocopa) and Osmia (Tergosmia) species), tightly fold the petal already on the flower (fig. 26) before it is transported to the nest in the mandibles and eventually unfolded inside the brood cell.
Comparative Biology of Ozbekosmia and Tergosmia
Apart from the three-layered structure of the brood cells common to both Ozbekosmia and Tergosmia, Osmia avosetta and the three species of Tergosmia differ in several aspects of their nesting biology (table 1). Osmia (Tergosmia) rhodoensis and O. (Tergosmia) tergestensis place their brood cells singly or in small groups of up to five cells in holes and fissures of rocks (fig. 22), between stones (figs. 23, 24), or in dense grass tussocks (fig. 25) (Ferton, 1897; Müller et al., 1997; C. Praz and C. Sedivy, personal obs.). The cells of both species, whose orientation varies from horizontal to nearly vertical, lie freely in these cavities and are neither glued to the substrate nor to each other. In contrast, O. (Tergosmia) lunata excavates a short (ca. 1.5 cm), more or less vertical burrow for each cell in rather hard soil (A. Müller, personal obs.; fig. 27). Unlike O. avosetta, whose cells are built a few cm below ground, cells of O. lunata reach the soil surface (fig. 27). Up to 10 cells are built immediately beside each other, separated by few mm only (fig. 29), and each cell is hidden under a thin and loose layer of small pebbles and earth fragments after its closure. Earth fragments are occasionally also used by O. tergestensis in narrow cavities to barricade the empty space in front of the cells (Ferton, 1897). Unfortunately, we do not know whether the excavated burrows of O. avosetta are actively filled or blocked with soil or other particles after completion of the brood cell(s).
Fig. 22
Two brood cells of Osmia (Tergosmia) rhodoensis in rock cavity; overlying stone removed to make cells visible (Jordan, Jerash, 23.4.2007; photo C. Sedivy). Figures 23, 24. Brood cells of O. (T.) tergestensis in cavities between stones; in both cases, overlying stone removed to make the cell visible. Petals used for brood cell construction were from Geranium and Helianthemum (fig. 23) and Geranium only (fig. 24) (Switzerland, Zeneggen, 10.7.1990; Photos A. Müller).

Fig. 25
Four closed brood cells and one cell still being provisioned of Osmia (Tergosmia) tergestensis between blades of dense grass tussock; grass blades removed to make cells visible (Italy, Gimillan (Aosta), 15.7.1996; photo A. Müller). Figure 26. Female of O. (T.) tergestensis biting off petal from flower of Geranium pyrenaicum Burm. fil. Petal is tightly folded before being transported to nest. On each visit, female removed one petal (Italy, Gimillan (Aosta), 15.7.1996; photo A. Krebs).

Fig. 27
Open brood cell of Osmia lunata in a short cavity excavated in rather hard ground; twig concealing nest entrance removed to make cell opening visible. Figure 28. Flower of Helianthemum sp. with one petal bitten off by female of O. (T.) lunata (Morocco, Tafraoute, 20.4.2009; photo A. Müller). Figure 29. Nest of O. (T.) lunata consisting of 10 brood cells at base of small shrub. Uppermost part of cell still under construction (top right) was the only visible sign of nest before excavation; the nine already closed cells were all hidden under thin layer of soil (Morocco, Tafraoute, 20.4.2009; photos A. Müller).

Figs. 30, 31
Diagrams of eggs of Osmia avosetta and of parasite thought to be Sapyga pulcherrima, lateral views, respectively, to same scale; anterior ends at right.

Figs. 32–36
Diagram of fifth larval instar of Osmia avosetta. 32. Entire larva, lateral view; rectangle identifies approximate area on abdominal segment 8 pictured in fig. 41. 33, 34. Head, frontal and lateral views, respectively; ptp = position of posterior tentorial pit. 35, 36. Right mandible, dorsal and outer views, respectively.

Figs. 37–40
SEM micrographs of fifth larval instar of Osmia avosetta. 37. Head, approximate frontal view. 38. Seta from area laterad of parietal band as identified by rectangle in figure 37 (but more lateral in view). 39. Right antenna showing three apical sensilla. 40. Close-up of left side of salivary opening, showing parallel ridges on upper inner surface.

Figs. 41–44
Microphotographs of fifth larval instar of Osmia avosetta. 41. Setae of pleural swelling, left side of abdominal segment 8, as identified by rectangle in figure 32. 42. Spiracle showing narrow peritreme relative to large atrial opening and wrinkled atrial wall. 43, 44. Short and long subatria, respectively. (Photos J.G. Rozen).

Table 1
Biological Characters of Osmia (Ozbekosmia) avosetta and Three O. (Tergosmia) Species (For references see text.)

Neither Turkish Osmia avosetta nor the Tergosmia species appear specialized with respect to the flowers exploited as petal sources. Known petal sources are Geranium and Linum for O. rhodoensis (Warncke, 1988; C. Praz and C. Sedivy, personal obs.) and Geranium, Helianthemum, Hieracium, and Ononis for O. tergestensis (Ferton, 1897; Benoist, 1931; Müller et al., 1997; figs. 23, 24, 26). Although the only petal sources recorded so far are Helianthemum for O. lunata (A. Müller, personal obs.; figs. 28, 29) and Onobrychis and Hedysarum for O. avosetta (this study), the colorful brood cells testify that both species use additional plant taxa as petal sources. However, the fact that all five brood cells of O. avosetta detected at the Iranian site were exclusively built with petals of Hedysarum indicates that the Iranian population might be specialized on a local scale.
Unfortunately, the phylogenetic relationships among species of the putative clade (Ozbekosmia, Tergosmia) are not yet resolved. Therefore, any hypothesis on the evolution of nesting biology within this clade appears to be premature. However, based on morphology (Warncke, 1988), there is little doubt that Osmia rhodoensis and O. tergestensis are close relatives. Further, O. lunata might be sister to a clade composed of O. avosetta and the other Tergosmia species due to (1) some possibly plesiomorphic characters compared to the other species (e.g., form of hind leg spur and of female clypeus) or (2) the structure of its cells being less distinctly three-layered in that the outermost layer is composed of petals cemented together with mud instead of consisting solely of petals. If in future phylogenetic analyses O. lunata should indeed turn out to be the sister species of the clade (avosetta (rhodoensis, tergestensis)), the evolution of nesting behavior within the clade (Ozbekosmia, Tergosmia) might have led from excavation of burrows in soil (O. lunata, O. avosetta) to the utilization of preexisting cavities (O. rhodoensis, O. tergestensis). This hypothesis is supported by the fact that species of the subgenus Hemiosmia, which is the possible sister taxon of the clade (Ozbekosmia, Tergosmia) (Praz et al., 2008), place their brood cells consisting of finely masticated leaves in excavated burrows in loose soil (Haeseler, 2008; C. Praz and C. Sedivy, personal obs. for Osmia difficilis Morawitz [Iran, Yasouj region, Road to Ghalat, June 1, 2009, new species record for Iran]). Under this evolutionary scenario, the central mud layer, which makes the brood cells firm and rigid, might possibly be interpreted as a preadaptation that later enabled the utilization of larger cavities as nesting sites, in which the cells have no contiguous support from the surrounding substrate. Interestingly, Hoplitis (Anthocopa) graeca and H. (Anthocopa) villosa, which are exceptions within Anthocopa in that both species use mud to cement together the petals, often use rather large cavities to place their brood cells as well (Friese, 1923; Petit, 1970; Westrich, 1990; A. Müller, personal obs.). In contrast, the other Anthocopa species accommodate their brood cells preferentially in narrow burrows in the ground (Mülleret al., unpubl.). The quantity of mud used by H. villosa to cement together petals is known to vary much, the extremes being pure petal cells on the one hand and mud cells with petals only on the outside on the other hand (Petit, 1970; Westrich, 1990). Unfortunately, it is not known whether the use of low mud quantities correlates with narrow cavities that give the brood cells all-embracing support.
One of the anonymous outside reviewers (C.D. Michener) kindly pointed out in his review the following: “Another group of bees that make cells somewhat similar to those of Osmia (Ozbekozmia) and its relatives is Megachile Group 1 of Michener (2007). Although these are in another tribe, the Megachilini, they appear to have evolved some similar behaviors. Although very well known as cutters of large leaf pieces for cell construction, some species use petals. Of particular interest in the present context are those of the subgenus Chrysosarus that construct cells of two layers of leaves or of petals with an intervening layer of clay, much as in O. avosetta. A brief summary with references to the original sources is by Michener (2007).” Zillikens and Steiner (2004) point out that Megachile (Chrysosarus) pseudanthidioides Moure from Brazil constructs an inner cell lining of petals and an outer one of leaves with a mud layer between. They state “The petals were not cut to the same size as the leaves, but were much larger and folded in the apical part of the chamber to form the cell closure.” Hence, they diagram a cell with the leaves of the outer layer cut to fit the diameter of the upper end of the cell, but the inner petal layer is folded inward from the sides. Thus, exemplars of two tribes have independently invented not only the use of petals in cell constructions but also a similar method of use.
All of the following species of Ozbekosmia and Tergosmia show close affinities to Fabaceae as pollen hosts (table 1). Osmia avosetta appears to be strictly specialized to the tribe Hedysareae (this study), Osmia lunata and O. tergestensis are more broadly oligolectic on Fabaceae (Benoist, 1931; A. Müller, unpubl.), and O. rhodoensis is polylectic with a preference for Fabaceae (A. Müller, unpubl.). Assuming the phylogenetic relationships within the clade (Ozbekosmia, Tergosmia) as hypothesized above, the polylectic habit of O. rhodoensis appears to be derived, supporting the growing evidence that polylectic species have often evolved from oligolectic ancestors while retaining the original pollen host, (Müller, 1996; Larkin et al., 2008; Sedivy et al., 2008), i.e., O. rhodoensis continues to collect pollen of Fabaceae but has added new pollen hosts. That pollen specialization on Fabaceae might be ancestral in this clade is further corroborated by the pollen preferences of species of the subgenus Hemiosmia, the possible sister taxon of the clade (Ozbekosmia, Tergosmia) (Praz et al., 2008). All Hemiosmia species investigated to date appear to be oligolectic on Fabaceae (Haeseler, 2008; A. Müller, unpubl.).
Acknowledgments
J.G.R. and J.S.A. extend their thanks to Robert G. Goelet, Chairman Emeritus, Board of Trustees, American Museum of Natural History (AMNH), for the support that permitted them to undertake this investigation. They also express their appreciation for the hospitality and guidance afforded by H.Ö. while undertaking fieldwork in Turkey.
Margaret A. Rozen took the SEM micrographs of the fifth instar with the assistance of Rebecca Rudolph and Mathew Frenkel, Microscopy and Imaging Facility, AMNH. Steve Thurston, Division of Invertebrate Zoology, AMNH, expertly organized and labeled all illustrative material.
C.S. thanks Silvia Dorn and the Walter Hochstrasser-Stiftung, ETH Zurich, for financing his trip to Iran. A.M. expresses his gratitude to Fritz Gusenleitner (Oberösterreichisches Landesmuseum Linz) for loaning pollen-loaded females of Osmia avosetta for the pollen-analytical studies, Albert Krebs for providing a picture of O. tergestensis, and Michael Widmer for support in the fieldwork on the nesting behavior of O. lunata. A.M. extends his appreciation to Azizollah Jaafari for his help in identifying the host plant species, to Shahrzad Azhari for her hospitality during the Iranian team's stay at Yasouj, and to Mostafa Salahi and Hadi Adelzadeh for their field assistance.
Josef Gusenleitner kindly identified specimens of Sapyga pulcherrima. Max Schwarz contributed valuable literature used in this study.
All authors extend thanks to the two anonymous outside reviewers for their helpful comments on improving the manuscript.